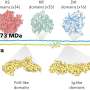
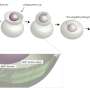
Orientation of protein patterns

During embryogenesis in the nematode Caenorhabditis elegans, the first cell division occurs transverse to the long axis of the fertilized egg. In a new study, biophysicists at LMU have now shown how this axis is reliably selected.
Many fundamental processes in biology depend on the formation of patterned distributions of specific proteins within cells. Examples include the localization of the cleavage plane prior to cell division and the direction of tissue growth. One paradigmatic example of cell polarization is the first cell division during embryogenesis in the nematode worm Caenorhabditis elegans, a well-established experimental model organism in developmental biology. In this system, the location of cell division is firstly determined by the so called PAR proteins (partitioning defective proteins). Since it is essential for the subsequent course of development, this operation must be tightly controlled. LMU biophysicist Erwin Frey and his research group have now identified two crucial mechanisms that contribute to the robust orientation of the polarized pattern of the PAR proteins along the long axis of the fertilized egg. The new findings appear in the online journal Nature Communications.
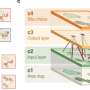
The first cell division of the elongated C. elegans egg is asymmetric, and determines the future front and back side of the worm. This anterior-posterior polarity of the embryo is defined by membrane-binding PAR proteins, which can be divided in two subgroups, aPARs and pPARs, binding to the anterior and posterior, respectively. In the unfertilized egg, all PAR proteins diffuse freely and are evenly distributed throughout the cell. In principle, all of them could bind to the membrane at any point. Due to a mutual antagonism between aPARs and pPARs each group of proteins soon builds a domain on the membrane excluding the other group from the membrane. "We use mathematical models and numerical simulations to explore the mechanisms that underlie the formation of protein patterns in cells. In this study, we analyzed what determines the orientation of the aPAR-pPAR pattern, specifically, what aligns the polarized pattern with the long axis of the egg," says Raphaela Gessele, a Ph.D. student in Frey's team and lead author of the paper.
Each group of PARs can bind to the membrane and antagonistically remove the other protein group from the membrane by phosphorylation. Among the aPARs A1 acts as a linker for A2 to collocate at the membrane. In the cytosol, the phosphorylated proteins are dephosphorylated only after some delay, enabling them to diffuse away from the detachment point before binding again. Simulations showed that the dynamics of cycling between the phosphorylated and dephosphorylated states of the aPARs and pPARs are a critical factor in the process of axis selection at patterning onset. Thus, the average duration of the delay between dissociation from the membrane and recovery of binding affinity is a vital parameter in determining the first orientation of the protein pattern along the long axis.
The study shows that the ellipsoidal geometry of the fertilized egg influences the patterning process. Owing to the difference in curvature, recently detached and freely diffusing proteins are more likely to reencounter the membrane at the poles than elsewhere in the cell. "Antagonistic proteins exclude each other proportionally to their membrane concentration which stabilizes domains. Depending on the respective lifetimes of the phosphorylated forms, the protein domains localize in the midcell region or at the cell poles at first; however, the final polarization in the three dimensional embryo is always aligned with the long axis," says Gessele.
Selection of the long axis polarization in the ellipsoidal embryo is further favored by the fact that the area of the transition zone between aPAR and pPAR domains, in which the two groups of proteins mutually exclude each other from the membrane, is minimized, reducing energetic costs. Owing to the elongated geometry of the fertilized egg, this is best achieved if each domain is confined to one pole. "Cell polarization plays a crucial role in very many biological systems," Frey points out. "Our results yield new insights into the mechanisms that cells employ to regulate this fundamental process."
More information: Raphaela Geßele et al. Geometric cues stabilise long-axis polarisation of PAR protein patterns in C. elegans, Nature Communications (2020). DOI: 10.1038/s41467-020-14317-w
Journal information: Nature Communications
Provided by Ludwig Maximilian University of Munich