How cells get moving
Archaea, bacteria and eukaryotes are what biologists call the three domains of life. Of these three, archaea form an important link within the evolutionary theory. They are the direct ancestors of eukaryotes, but resemble bacteria in structure and organization. Archaea can colonize hot sulphur springs or extremely saline lakes, but can also be found in the ocean or in the human intestine and on the skin. Unlike bacteria, archaea have been relatively little researched—because no pathogenic forms have been identified so far. A research team led by Professor Sonja-Verena Albers from the Professorship of Microbiology at the University of Freiburg and Professor John Tainer from the University of Texas in Austin, U.S., has now succeeded in identifying proteins that are essential for the assembly of the motility structure, i.e. for the ability of archaea to move actively—and hence also for the locomotion of cells. The team has presented its findings in the latest Nature Microbiology.
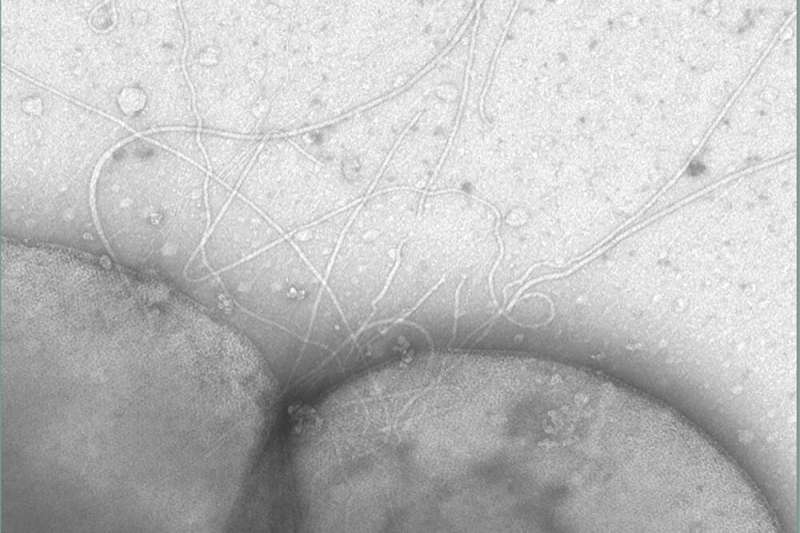
It is important for microorganisms to be able to move actively—so that if their environment deteriorates they can seek better living conditions. Bacteria use what is known as the flagellum, a complex structure requiring up to 50 proteins that assemble according to a strict timetable. Scientists assumed that archaea used the same structure as bacteria to "swim" from one place to another. But after sequencing the first archaeal genomes, the researchers discovered that archaea did not possess flagella operons. Instead, archaea swim using a structure called an archaellum. It consists of only seven subunits in the model organism Sulfolobus acidocaldarius used by Albers, which lives in highly acidic hydrothermal springs. "Nevertheless, this relatively simple structure can perform the same functions as the bacterial flagellum," she explains.
She and her team have already discovered the structure of the protein FlaI required for this and have shown that it forms the motor complex of the archaellum in combination with the proteins FlaX and FlaH. The Freiburg scientists also described the protein FlaF, which bonds with the model organism's only cell wall protein and anchors it there. "This enables archaea to interact with the environment—and thus with human cells," explains Albers. In the current study, the researchers show that the protein FlaG is able to form a filament which is like a fiber. Together with FlaF, FlaG then forms a protein complex that is essential for the assembly of the archaellum. This complex is probably located at the tip of the FlaG filament, which enables FlaG and FlaF to interact with the cell wall.
In addition, the scientists proved that cells without a cell wall can form an archaellum but cannot swim. Albers says that this suggests that the interaction between FlaG and FlaF and the cell wall contributes to the function of the archaellum. "We showed that FlaG and FlaF are essential for the rotation of the archaellum and hence for the locomotion of the cell," she says.
More information: Chi-Lin Tsai et al. The structure of the periplasmic FlaG–FlaF complex and its essential role for archaellar swimming motility, Nature Microbiology (2019). DOI: 10.1038/s41564-019-0622-3
Journal information: Nature Microbiology
Provided by University of Freiburg