Researchers develop micro-electro-fluidic probe (MeFP) to isolate and pattern cells
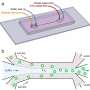
A team of researchers from NYU Abu Dhabi have developed a dielectrophoresis (DEP) enabled MicroelectroFluidic Probe (MeFP) that has the ability to sequentially separate and pattern mammalian cells in an open microfluidic system. Mammalian cells are tiny (approximately one tenth of a single hair strand diameter) and therefore becomes very challenging to selectively enrich and pattern cells at the single cell resolution. The developed tool is capable of selectively enriching cells of interest in a magnetic-like arrest that leads to grabbing target cells from the fluid stream, while leaving non-target ones within the flow untouched. Consequently, arrested cells get released and patterned on substrate in a 2-D-printing-like process.
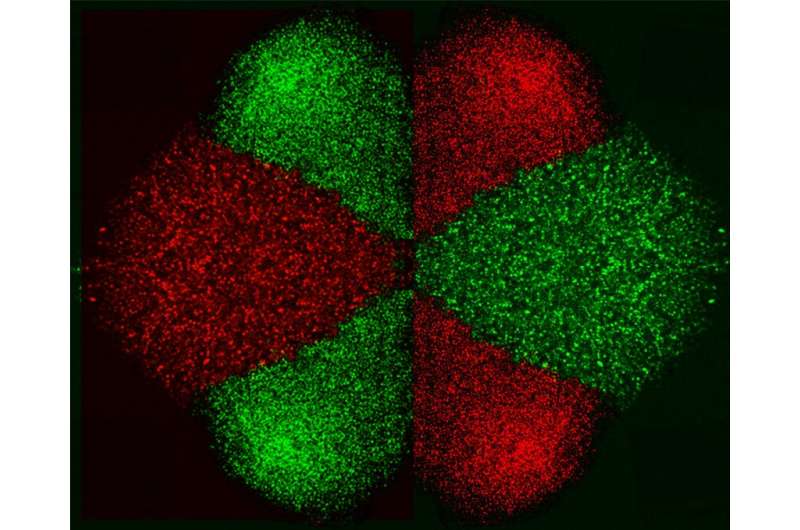
The study demonstrated the MeFP with up to 100% isolation efficiency, 90% separation purity, and a pattern deposition rate of a few seconds. Using this method to sort, purify, and assemble cells into controlled patterns is the first step in the tissue engineering process.
The device also has a potential application in the sequential capture and characterization of circulating tumor cells (malignant cells found in cancer patients' blood). Since the MeFP can directly deposit captured cells on any bottom flat substrate, these cells can be subsequently tested with multiple chemotherapeutic drugs using only the microfluidic feature of the same device.
In the paper, "Microelectrofluidic Probe for Sequential Cell Separation and Patterning," published in the journal Lab on a Chip, the researchers present the process of creating a tool that can separate and pattern cells using DEP forces within an open "channel-less" microfluidic system, allowing cellular patterns with complex tissue-like structure to grow. The MeFP is a microfluidic probe with injection and aspiration apertures, integrated with an array of micro-hump electrodes on its tip. By adjusting the flow configuration of the MeFP, the researchers were able to pattern a cell culture containing two different types of cells for homotypic and heterotypic cell interaction studies.
With the MeFP, biological cells of different types can be sequentially sorted and simultaneously patterned, on any flat substrate, to form the required cell patterns and potentially tissue constructs. Examples of different cell types include cancer cells, stem cells, immune and red blood cells, to name a few.
"The MeFP is a multifunctional tool for manipulating cells in open channel-less space," said lead researcher and assistant professor of Mechanical and Biomedical Engineering at NYUAD Mohammad Qasaimeh, "this demonstration of its simple, dynamic and customizable nature will inspire new cell patterning, tissue engineering, and life science applications."
"The developed tool can be used to scan across any flat substrate, and can separate specific cells based on their electronic signatures,"' commented first author and Global Ph.D. Fellow in Mechanical Engineering at NYUAD Ayoola T. Brimmo.
"This is the first study that combines DEP forces within an open microfluidic probe, which possess the features of hydrodynamic confinements and scanning capabilities," said second author and senior research scientist in Mechanical and Biomedical Engineering at NYUAD Anoop Menachery.
More information: Ayoola T. Brimmo et al, Microelectrofluidic probe for sequential cell separation and patterning, Lab on a Chip (2019). DOI: 10.1039/C9LC00748B
Journal information: Lab on a Chip
Provided by New York University