The topology of disordered 3-D graphenes: Rosalind Franklin's pre-DNA problem untangled
Disordered three-dimensional graphenes are carbon materials present in batteries, water filters, gas masks, high-temperature ceramics, electrochemical sensors and insulation. They also have more specialised uses, such as protecting the Parker solar probe spacecraft from burning up on its approach to the sun.
Rosalind Franklin, the scientist who would later deduce the helical geometry of DNA, first discovered this class of materials in 1951. Most carbon-containing materials develop small layered regions of graphene when heated. Upon further heating, to thousands of degrees, she found (to her surprise) a complete reluctance of the carbons to convert to the most stable form of carbon graphite, making it supremely metastable.
Explanations for this reluctance to graphitize have centered around either crosslinks within the structure, knotted ribbon-like structures or warping of the sheets into bowl- or saddle-shaped geometries. However, experiments were unable to resolve and combine these suggestions into a coherent model of the nanostructure.
Researchers from Curtin University, Australia and the University of Cambridge have now published a possible solution to Franklin's problem in Physical Review Letters. They turned to large scale simulations using Australia's Pawsey supercomputer to self-assemble the largest and most accurate networks of disordered 3-D graphenes to date.
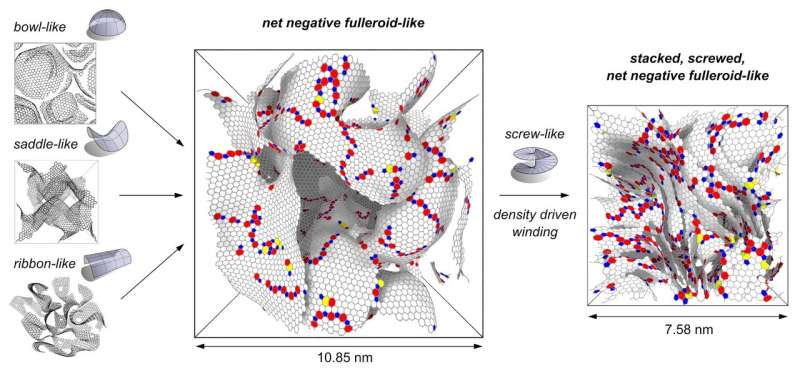
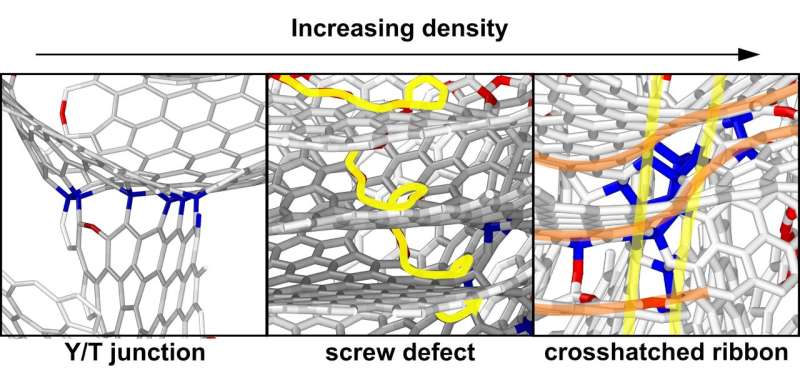
They developed a new measure for the global curvature of the networks and found that for all densities, excess saddle-shaped graphene sheets are present. These saddle shapes are caused by the integration of 7- or 8-membered rings within the hexagonal graphene network. This warping allows it to connect in 3-D and the researchers suggest it is the cause of the material's resistance to convert into graphite.
How about Franklin's small regions of layered graphene? The researchers found that upon increasing the density of the material, the graphene sheets wound up like a spiral staircase. This screw or helix defect is well known in graphite but has not been suggested in these disordered materials. A variety of other defects were discovered, which resolve many issues of the graphene network being both curved and layered.
These results open up possibilities for understanding and engineering carbon materials for applications in supercapacitors, carbon fibers and high-temperature ceramics applications. However, more work is needed to experimentally confirm some aspects of the model.
In terms of new applications, the researchers suggest that carbon materials could be topologically tuned and optimized for a given product. For example, and of particular industrial importance to making batteries and electrodes, disordered carbons could be transformed into graphite (instead of relying on unsustainable mining practices).
-
The Curtin Carbon group visualising the large scale carbon networks using the Curtin HIVE immersive display. Credit: twitter.com/CurtinHIVE/status/999853141267890180?s=20 -

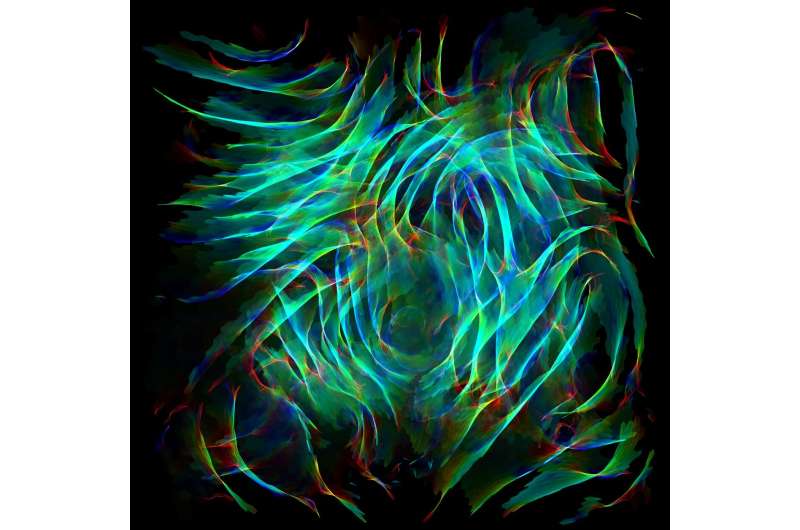
This shows angular defects, a measure of curvature, on a disordered 3D graphene surface. Red for negative and blue for positive curvature. Credit: Jacob Martin
There is a pleasing connection with Franklin's later work on DNA in that the solution to her earlier problem of non-graphitizability in carbon materials could also lie in topology and the famed helix structure.
More information: Topology of Disordered 3D Graphene Networks, JW Martin, C de Tomas, I Suarez-Martinez, M Kraft and N Marks, Physical Review Letters, 2019. DOI: 10.1103/PhysRevLett.123.116105
Journal information: Physical Review Letters