Next generation synthetic covalent 2-D materials unveiled
A team of researchers at the National Graphene Institute at The University of Manchester have developed a new method to synthesize 2-D materials that are thought to be impossible or, at least, unobtainable by current technologies.
Graphene was the world's first two-dimensional material, which subsequently opened the gates for the isolation of other two-dimensional materials.
Graphene and other 2-D materials usually have a 3-D counterpart known as a 'bulk analogue'. For example graphene is a single layer of carbon atoms derived from graphite.
Recently, there has been a growing interest in the fabrication of synthetic 2-D materials that have no layered bulk analogue. Researchers have started to look at 2-D materials that do not have a 3-D counterpart.
Traditionally, 2-D materials are isolated by a process called mechanical exfoliation –taking the bulk material and exfoliating the layers from each other until a single layer is achieved.
In contrast to these layered crystals, those materials that have no layered structures are held together by covalent bonds between the atomic planes, which do not allow mechanical exfoliation.
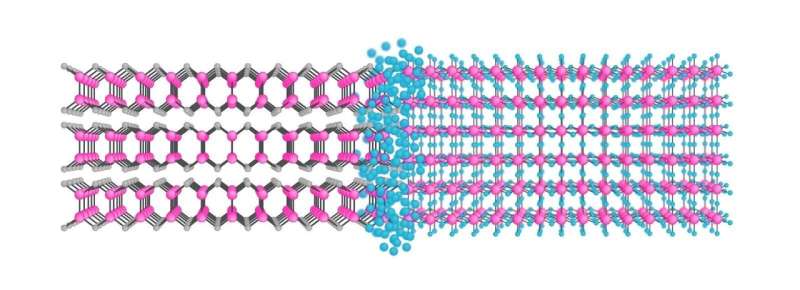
As published in Nano Letters, by using chemical conversion, the team at the University were able to convert layers of existing layered materials into a new covalent two-dimensional material. As an example, mechanically exfoliated 2-D indium selenide (InSe) is converted into atomically thin indium fluoride (InF3), which has a non-layered structure and therefore cannot be possibly obtained by exfoliation, by a fluorination process.
Effectively, the proposed chemical conversion strategy of 2-D material is nothing but sewing atomic layers of existing 2-D materials together by chemical modification.
The obtained new 2-D indium fluoride is a semiconductor, exhibiting high optical transparency across the visible and infrared spectral ranges and could potentially use as a 2-D glass.
Professor Rahul Nair at the National Graphene Institute and Department of Chemical Engineering and Analytical Science who led the team said: "Chemical modification of materials has proven to be a powerful tool for obtaining novel materials with desired and often unusual properties. There is still further work to be carried out to understand chemical conversion of 2-D materials at the atomic scale, including effects of relative orientation and synergy between individual atomic layers on their chemical reactivity. We believe our work provides a significant advance in materials science and is a clear milestone in the development of artificial 2-D materials."
Vishnu Sreepal, who led the experiments and the lead author of this paper said "Our work clearly demonstrates the possibility of creating artificial 2-D covalent materials. The process is controllable, easy to execute and very effective. By precisely controlling the thickness of the starting 2-D layers the thickness of the new covalent 2-D materials can be controlled with an atomic-scale precision. The new covalent 2-D material can also be controllably doped with dopants".
"We also demonstrate the scalability of our approach by chemical conversion of large-area, thin InSe films into InF3 films."In addition, the team envisages that such chemical conversion can be extended to van der Waals heterostructures to obtain artificial hetero covalent solids.
By layering atoms in a precisely chosen sequence known as heterostructures, designer materials with certain characteristics can be created that don't occur naturally and offer specified qualities. Researchers assemble these new materials in sequences relevant to their intended application, in a process similar to stacking Lego bricks. By demonstrating the possibility of 2-D covalent solids, researchers now have more 'legos' in their playground to create novel materials with custom made properties.
More information: Vishnu Sreepal et al. Two-dimensional covalent crystals by chemical conversion of thin van der Waals materials, Nano Letters (2019). DOI: 10.1021/acs.nanolett.9b02700
Journal information: Nano Letters
Provided by University of Manchester