Biologists design a plug-and-play controller for gene expression
Researchers from two University of Texas System institutions have engineered biological components that can rewire genetic response pathways, creating a template for designing new cellular behaviors with wide-ranging potential applications.
Dr. Faruck Morcos of The University of Texas at Dallas and Dr. Clement Chan of UT Tyler have collaborated on modular versions of repressors—the proteins that block genetic instructions.
In a paper recently published in Nucleic Acids Research, they proposed a new strategy to create living cells that can sense multiple signals via reporter proteins. Their work, which has potential applications in medical diagnostics, originates from an interdisciplinary field called synthetic biology.
"The goal of synthetic biology is to use existing biological systems to develop solutions for problems," said Morcos, an assistant professor of biological sciences at UT Dallas. "It's an engineering approach to biology."
The research team aims to build diagnostic cells that would inhabit a patient's body and detect a variety of symptoms. After the symptoms have subsided, the engineered cells can be switched off simultaneously in response to a specific "reset" signal so that they can monitor if the symptoms reoccur.
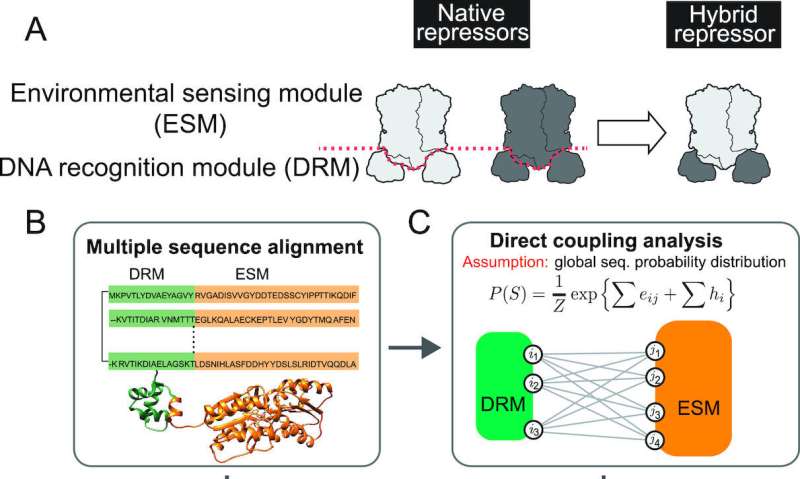
These living diagnostics were developed based on the cellular machinery for regulating gene expression. In living cells, genes hold instructions for making the proteins that carry out various processes. Those instructions are copied in a process called transcription. Signals from the environment or other cells activate these copiers. The segments of a DNA strand where transcription begins are called promoters.
In some situations, a repressor agent can attach to a promoter, blocking a genetic segment from being copied and overriding its instruction. Due to the manner in which evolution has occurred, each repressor is extremely specific in purpose.
"One specific signal controls whether or not a specific gene gets expressed," said Chan, an assistant professor of biology and biochemistry at UT Tyler. "These interactions are so individualized that it has been difficult to engineer a tool that either uses one signal to control gene expression by multiple promoters or uses multiple signals to control the same promoter. This rigidity poses a major challenge in creating new cellular behavior."
The research team engineered repressors to solve this problem. Their synthetic repressor molecules consist of distinct, modular protein segments for signal detection and promoter recognition that can be separated and recombined with other repressor segments. Like a universal remote that can send various signals to multiple devices, swapping segments changes the target that will be blocked.
But randomly pairing protein segments would be like trying to use the remote on hardware from all manufacturers—compatibility is uncertain, although your TV is more likely to respond than your microwave. So the researchers developed a computer modeling program to predict which pairings should be fruitful.
"Some of our hybrid repressors were more functional than others," Morcos said, "so we set out to understand the molecular basis of compatibility."
"If we can figure out why certain pairings aren't compatible, we could learn how to modify them to make them work," Chan added.
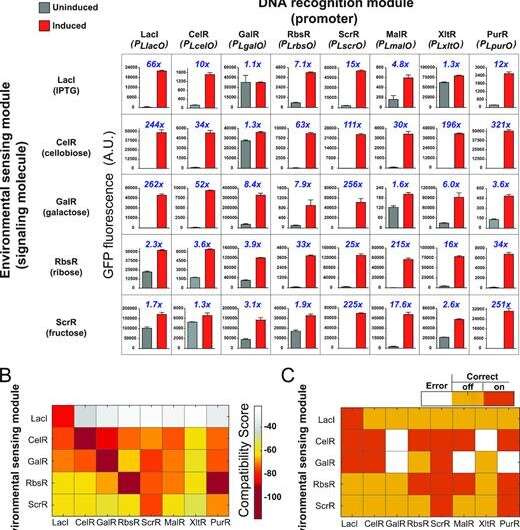
The team can predict with 87 percent accuracy whether a promoter-signaler pairing will work.
"Our algorithm finds things that these related protein segments have in common, features that have been fine-tuned through evolution. These features indicate a compatibility score," Morcos said. "That was phase one of the study—building this kind of biological circuit that, to our knowledge, has never been built before."
The group of repressor proteins being used, called the LacI family, has more than 70,000 known variations—but only a handful have been characterized. This repressor family was chosen because of its frequent use in synthetic biology and because a previous study involving Chan identified its distinct signal detection and promoter-recognition modules.
"LacI family members are used across many species to control gene expression by sensing signals within the cell and in the environment," Morcos said.
UT Dallas doctoral student Xianli Jiang led the computational effort to model modular compatibility, while Rey P. Dimas, a technician at Chan's lab who joined the project while an undergraduate at UT Tyler, worked on building and testing these hybrids experimentally.
"We don't know the signals that each Lacl family member can detect," Morcos said. "So we are creating a library of interactions, and as more of these LacI repressors are characterized, our knowledge base grows."
Jiang explained the library's usefulness with an example of a researcher who has chosen a gene to repress, and knows its promoter.
"This library is a tool for all researchers," Jiang said. "They can use our compatibility model to predict if any proteins they engineer will block transcription or not. Or they can use the protein segments we've already designed."
The second stage of the study demonstrated how this technique could engineer the behavior of a cell for beneficial purposes: living diagnostic cells that can measure multiple physiological parameters.
This system would employ what the researchers describe as a "toggle switch" to indicate it has received a signal for a positive result—high blood sugar, for example. The switch would then give instructions to turn on a fluorescent light.
"The switch controls the expression of a fluorescent reporter gene," Chan said. "We can build multiples of these switches, one for each parameter—presence of a pathogen, blood glucose level, whatever."
Such a system also would use a "master reset" signal to shut off all the lights so another reading can be taken later.
"In a home alarm system, multiple signals trigger different alarms—at the front door, a window, the garage, and so on," Morcos said. "Each alarm can turn on independently, but from your control panel, one code will turn everything off."
More information: Rey P Dimas et al. Engineering repressors with coevolutionary cues facilitates toggle switches with a master reset, Nucleic Acids Research (2019). DOI: 10.1093/nar/gkz280
Journal information: Nucleic Acids Research
Provided by University of Texas at Dallas