Artificial membrane binding proteins to direct stem cells to the myocardium
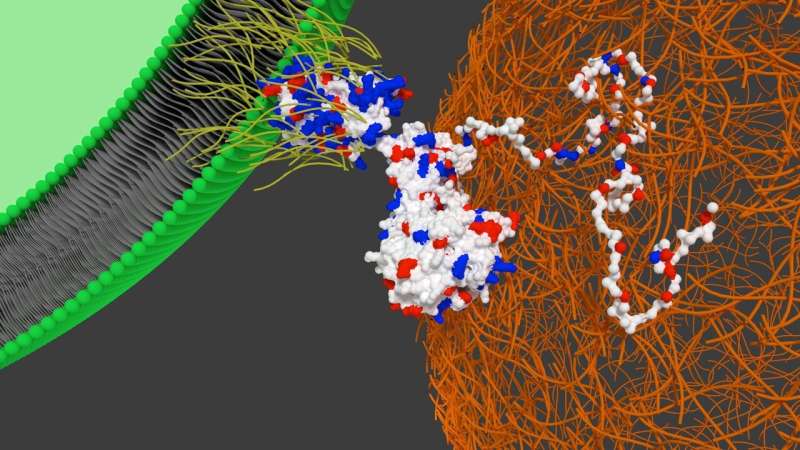
![Fig. 1 The artificial plasma membrane binding construct. (a) Schematic showing the interaction of the polymer surfactant conjugated supercharged fusion [CshA_scGFP][S] construct with the bilayer of the cytoplasmic membrane. The structure shows the surface charge potential, highlighting the high positive charge (blue) on the supercharged GFP domain and the negative charge (red) on the CshA globular domain, as well as the “catch” and “clamp” domains of CshA. (b) The structure of the anionic polymer surfactant glycolic acid ethoxylate 4-nonylphenyl ether (S), which electrostatically complexes with the supercharged GFP moiety. Credit: Chemical Science, open access: https://pubs.rsc.org/en/content/articlelanding/2019/sc/c9sc02650a#!divAbstract Artificial membrane binding proteins to direct stem cells to the myocardium](https://scx1.b-cdn.net/csz/news/800a/2019/artificialme.jpg)
In a world first, scientists have found a new way to direct stem cells to heart tissue. The findings, led by researchers at the University of Bristol and published in Chemical Science, could radically improve the treatment for cardiovascular disease, which causes more than a quarter of all deaths in the UK.
To date, trials using stem cells, which are taken and grown from the patient or donor and injected into the patient's heart to regenerate damaged tissue, have produced promising results.
However, while these next generation cell therapies are on the horizon, significant challenges associated with the distribution of the stem cells have remained. High blood flow in the heart combined with various 'tissue sinks," that circulating cells come into contact with, means the majority of the stem cells end up in the lungs and spleen.
Now, researchers from Bristol's School of Cellular and Molecular Medicine have found a way to overcome this by modifying stem cells with a special protein so they 'home' to heart tissue.
Dr. Adam Perriman, the study's lead author, associate professor in Biomaterials, UKRI Future Leaders Fellow and founder of the cell therapy technology company CytoSeek, explained: "With regenerative cell therapies, where you are trying to treat someone after a heart attack, the cells rarely go to where you want them to go. Our aim is to use this technology to re-engineer the membrane of cells, so that when they're injected, they'll home to specific tissues of our choice.
"We know that some bacterial cells contain properties that enable them to detect and 'home' to diseased tissue. For example, the oral bacterial found in our mouths can occasionally cause strep throat. If it enters the blood stream it can 'home' to damaged tissue in the heart causing infective endocarditis. Our aim was to replicate the homing ability of bacteria cells and apply it to stem cells."
The team developed the technology by looking at how bacterial cells use a protein called an adhesin to 'home' to heart tissue. Using this theory, the researchers were able to produce an artificial cell membrane binding version of the adhesin that could be 'painted' on the outside of the stem cells. In an animal model, the team were able to demonstrate that this new cell modification technique worked by directing stem cells to the heart in a mouse.
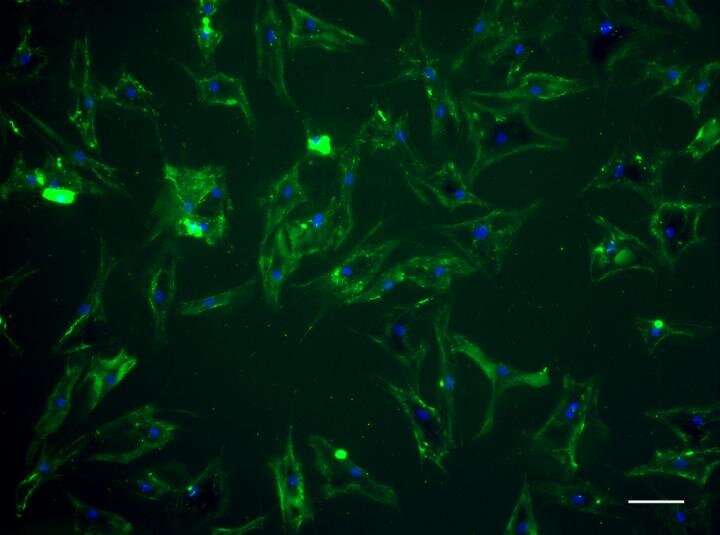
Dr. Perriman added: "Our findings demonstrate that the cardiac homing properties of infectious bacteria can be transferred to human stem cells. Significantly, we show in a mouse model that the designer adhesin protein spontaneously inserts into the plasma membrane of the stem cells with no cytotoxity, and then directs the modified cells to the heart after transplant. To our knowledge, this is the first time that the targeting properties of infectious bacteria have been transferred to mammalian cells.
"This new technique carries enormous potential for the seven million people currently living with heart disease in the UK."
More information: Wenjin Xiao et al. Designer artificial membrane binding proteins to direct stem cells to the myocardium, Chemical Science (2019). DOI: 10.1039/C9SC02650A
Journal information: Chemical Science
Provided by University of Bristol