Veritable powerhouses—even without DNA

Whether human beings or animals, plants or algae: the cells of most life forms contain special structures that are responsible for energy production. Referred to as mitochondria, they normally have their own genetic material, in addition to that found in the nucleus. Uwe John and colleagues at the Alfred Wegener Institute have now identified the first-ever exception to this rule in a single-celled parasite. The mitochondria of the dinoflagellate Amoebophrya ceratii appear to produce energy just like our own mitochondria, but without any genetic material, as the team reports in the journal Science Advances.
They are successful, diverse and practically ubiquitous: dinoflagellates make up a major portion of the plankton found in our oceans and have a broad range of lifestyles. Roughly half of the two thousand known species use photosynthesis like plants, while others are predatory or alternate between different forms of food, depending on what's available. And last but not least, the group also includes parasites. An international team from France, Canada, Egypt, UK,Czech Republic and Germany led by Uwe John, a biologist at the Alfred Wegener Institute, Helmholtz Centre for Polar and Marine Research (AWI), recently took a closer look at one of these parasites' genetic material—and were amazed by what they found.
The researchers originally discovered their object of study within the cells of other dinoflagellates from the genus Alexandrium, which includes numerous species that are infamous marine alchemists and tend to form toxic algal blooms. In some cases, entire carpets of these single-celled organisms cover the water's surface and produce saxitoxin, a neurotoxin that can be dangerous for humans. But there are also parasites that can keep these blooms in check, e.g. the species Amoebophrya ceratii, which is at the heart of the current study.
"These single-celled organisms float in the water as what we call dinospores until they find a suitable host," Uwe John explains. They then latch onto their victim, penetrate it and eat it from the inside out. In the process, they grow larger and larger, eventually reaching a stage with multiple nuclei. Like a worm, they then crawl out of the dead host and dissolve into hundreds of new dinospores. This infection cycle only takes three or four days, and can decimate Alexandrium populations. "In fact, it may be thanks to these parasites that toxic algal blooms are now less frequent off the coast of Brittany than just a few years ago," says John—an aspect that makes investigating the toxic-algae killers all the more exciting.
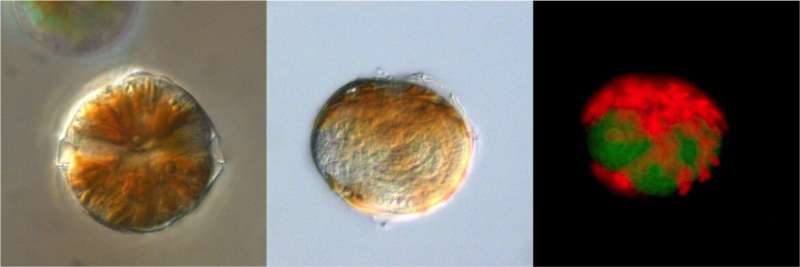
In this context, the team sequenced the genome (all the genetic material) of Amoebophrya, which consists of roughly 100 million base pairs. That's extremely low by dinoflagellate standards: other species have genome a thousand times larger, which is much larger than even the human genome. That being said, a small genome isn't exactly unusual for a parasite. Many of those pursuing this lifestyle don't produce all of the metabolic products they need to survive; they simply steal them from their host. Though this makes them host-dependent, it also means there are many genes they can simply do without. But that's not the road that Amoebophrya ceratii chose to go down. "In this species, nearly all metabolic processes still work, so it should also be able to get by quite well on its own," says Uwe John. And it can do so with significantly less genetic material than any other dinoflagellate.
This reduction is especially pronounced in that part of the genome that lies outside the nucleus. In plants and algae, their own DNA is not just found in the nucleus and mitochondria, but also in the plastids, which they need for photosynthesis. For dinoflagellates in general, this plastid DNA is fairly basic, and consists of only 14 genes. Yet Amoebophrya ceratii would appear to have done away with the plastids entirely, and with all but one of their genes.
Yet the 'budget cuts' that the parasite has pursued with regard to its mitochondria are even more impressive. In related species, these tiny power plants' DNA still contains three genes, which experts have generally assumed to be indispensable. But Amoebophrya ceratii has apparently cut out the mitochondria genome entirely; despite painstaking efforts, the team was unable to find any trace of it. Two of the three genes are nowhere to be found, and the third, cytochrome c oxidase 1 (COX1 or COI), has migrated to the nucleus. "That absolutely amazed me," says Uwe John, "because there's no other oxygen-breathing life-form on record that has no genetic material of its own in its mitochondria."
This downsizing could come in handy when the parasites need to rapidly form a large number of new dinospores. According to John, "Perhaps it's then more effective to regulate all processes using the nucleus. This is likely also the most efficient way to use the host's resources." But none of this would do any good if the energy supply broke down. Yet that seems unlikely: the mitochondria work quite well in all stages, even allowing the dinospores to swim rapidly when looking for a host. "Apparently, these parasites have found a strategy all their own when it comes to energy production," AWI researcher Uwe John summarises. "To produce energy, they require only part of the five known protein complexes found in the mitochondria of human beings and all animals."
The experts hope that their findings will help us understand the evolution of the dinoflagellates and their relatives. This would also be interesting because the latter include other parasites and the pathogens that cause diseases like malaria. In addition, the findings could offer new insights into the evolutionary histories of mitochondria and plastids: both were originally independent life forms, which were absorbed by other single-celled organisms in the primordial past, and lived on inside them as so-called endosymbionts. With the passage of time, their genetic material was gradually reduced and they essentially became 'service providers' that were no longer capable of surviving on their own. Yet Amoebophrya ceratii appears to have taken this development one step further, and taken away the last vestiges of its endosymbionts' genetic autonomy.
More information: U. John el al., "An aerobic eukaryotic parasite with functional mitochondria that likely lacks a mitochondrial genome," Science Advances (2019). DOI: 10.1126/sciadv.aav1110 , advances.sciencemag.org/content/5/4/eaav1110
Journal information: Science Advances
Provided by Alfred Wegener Institute
















