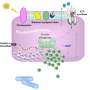
Key players in the marine nitrogen cycle use cyanate and urea
The ammonia oxidizing archaea, or Thaumarchaeota, are among the most abundant marine microorganisms. Yet, scientists are still discovering which factors allow them to thrive in the ocean. A research team from the Max Planck Institute for Marine Microbiology in Bremen and the University of Vienna was now able to show that marine Thaumarchaeota have a broader metabolism than previously thought. The results are published in the journal Nature Microbiology.
The Thaumarchaeota play a key role in the marine nitrogen cycle. They gain energy for growth by converting ammonia, which is the most reduced form of inorganic nitrogen, to a more oxidized form: nitrite. These so-called ammonia oxidizing archaea were discovered little more than a decade ago, yet these organisms make up a large part of the marine microbial community, thriving in the oceans despite ammonium being present only at very low concentrations.
Even though the Thaumarchaeota are such a key part of the marine nitrogen cycle, little is known about the physiology of these small and enigmatic microorganisms. In general, they are considered to be metabolically restricted, relying on ammonia as an energy source. A new study by Katharina Kitzinger and colleagues from the Max Planck Institute for Marine Microbiology in Bremen, Germany, the University of Vienna, Austria, the Georgia Institute for Technology, USA, the Carl von Ossietzky University Oldenburg, Germany, and the MARUM—Center for Marine Environmental Sciences in Bremen, Germany, now reveals that this is not quite true. Rather, the authors show that marine ammonia oxidizing archaea can also utilize organic nitrogen sources.
"We show for the first time that both environmental and cultured marine ammonia oxidizing archaea can use cyanate, a simple organic nitrogen compound, as an additional energy source," Kitzinger explains. Further, they show these microorganisms also use that urea, another organic nitrogen compound. These findings are important as cyanate and urea are common nitrogen and energy sources in the oceans. The Thaumarchaeota's ability to supplement their metabolism with these compounds might be one reason for their outstanding success in the oceans.

Kitzinger is especially intrigued by how the marine ammonia oxidizing archaea are able to use cyanate. "We still aren't sure exactly how they do it. They don't have the typical enzyme repertoire needed to use cyanate. It will be exciting to see which enzymes allow marine ammonia oxidizing archaea to use cyanate, if these organisms have an even larger metabolic versatility than we know now, and how this versatility shapes their ecology," says Kitzinger.
More information: Katharina Kitzinger et al, Cyanate and urea are substrates for nitrification by Thaumarchaeota in the marine environment, Nature Microbiology (2018). DOI: 10.1038/s41564-018-0316-2
Journal information: Nature Microbiology
Provided by Max Planck Society