July 31, 2015 report
Researchers find it is possible to use cyanate as an energy source for nitrifiers
An international team of researches has found an example of a nitrite oxidizing organism that can convert cyanate into ammonium and carbon dioxide. In their paper published in the journal Nature, the team describes their research which had both a genetic component and field experiments, and why they believe what they found might make the case for cyanate being classified as a key controller of the nitrification process. Lisa Stein of the University of Alberta offers a News & Views piece on the work done by the team, in the same journal edition.
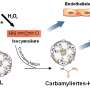
For many years, scientists have believed that microorganisms that serve as ammonia oxidizers drive the nitrification process, because it was believed they were the ones that created the food for nitrite oxidizing microorganisms. Now it appears that such thinking might have to be modified, as the researchers with this new effort have found an example of an ammonia-oxidizing thaumarchaeote (organisms of the Archaea phylum), Nitrososphaera gargensis1, that uses cyanate as the one and only source of energy and reductant—showing that nitrite oxidizers can provide the food for ammonia oxidizers. Thus it appears to go both ways.
The team made the discovery while conducting genome tests of nitrite oxidizers—they found genes that encode for cyanase, an enzyme that is known to break down cyanate, a sign that they likely could use cyanate as an energy source. To find out for sure, they conducted an aerobic growth of N. gargensis1 (an ammonia oxidizer) using just cyanate, proving that it could be done—it is now classified as the only organism able to carry out such a process.
The findings by the team are an important discovery because it will help scientists better understand the nitrogen cycle in general and the nitrification process more specifically—and that should help to better understand the issues involved in solving such problems as unabated nitrification that occurs due to over fertilization of croplands leading to pollution and dead zones in the sea. And as Stein, notes, we have entered a time where human production of fixed nitrogen is greater than that produced naturally with the result being a nitrogen cycle that is now unbalanced—suggesting, perhaps that leveraging organisms such as N. gargensis1 could help restore that balance.
More information: Nature (2015) DOI: 10.1038/nature14856
Journal information: Nature
© 2015 Phys.org