Traditional eutectic alloy brings new hope for high energy density metal-oxygen batteries
Current lithium-ion intercalation technology, even when fully developed, is insufficient to satisfy the increasing demand for high-energy-density power sources for electric vehicles and electronics. Thus, non-aqueous alkali metal-oxygen (AM-O2: AM = Li, Na, etc.) batteries are positioned to replace conventional lithium-ion batteries due to their ultrahigh theoretical energy density.
However, AM is extremely reactive toward air and almost all nonaqueous electrolytes, resulting in significant parasitic reactions. Furthermore, uncontrollable Li or Na metal plating/stripping, generally emerging as dendrites, easily induces short circuits accompanied by fire/explosion events. Therefore, to achieve a safe and stable AM-O2 cell, it is important to solve the dendrite and oxidation/corrosion issues.
Recently, a research team led by Zhang Xinbo from the Changchun Institute of Applied Chemistry (CIAC), Chinese Academy of Sciences, Yan Junmin from Jilin University, Zhang Yu from Beihang University Beijing developed a long-life AM-O2 battery using Li-Na eutectic alloy as novel metal anode for the first time. Their findings were published in Nature Chemistry.
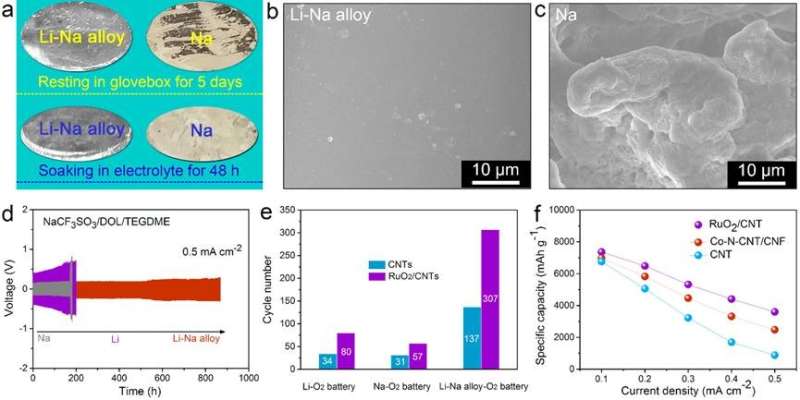
They found that Li and Na exhibited similar reaction activities and therefore both could be employed as active components in batteries without sacrificing the specific capacity compared with other alloys (e.g., Na-Sn alloy). In addition, alloying Li and Na improved the corrosion resistance and suppressed metal dendrite growth.
In a Li-Na alloy battery, with the help of the electrolyte additive, the resultant dendrite-suppressed, oxidation-resistant, and crack-free Li-Na alloy electrode endowed the newly proposed aprotic bimetallic Li-Na alloy-O2 battery with good performance.
Furthermore, by introducing efficient O2 reduction/evolution catalysts (e.g., Co/NCF), the cycling life and rate capability of Li-Na alloy-O2 battery were significantly improved.
"We believe that this strategy can also be applied to other metal electrodes, such as Zn, Mg, Ca, Al and so on," said Zhang.
Meanwhile, this study provides a guidance for developing other bimetal batteries such as bimetal ion batteries and bimetal-S batteries. These batteries have new chemistry, exhibit much better electrochemical performance than mono-metal batteries, and adopt collaborative methods to release the great potential of alkali metal anode.
More information: Jin-ling Ma et al, Prevention of dendrite growth and volume expansion to give high-performance aprotic bimetallic Li-Na alloy–O2 batteries, Nature Chemistry (2018). DOI: 10.1038/s41557-018-0166-9
Journal information: Nature Chemistry
Provided by Chinese Academy of Sciences