The pentagon and the bean—redrawing the structure of an immune system protein
Researchers have revealed the structure of an essential immune protein, creating future possibilities to develop more effective medicines for a range of illnesses from cancer to neurological diseases. University of Tokyo researchers made this discovery with computerized image analysis and modern electron microscope imaging.
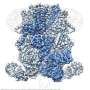
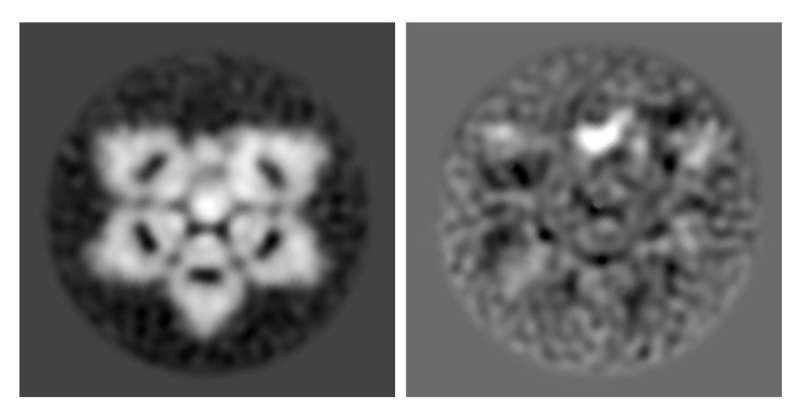
Researchers verified the structure of natural immunoglobulin M (IgM) protein, an important part of the immune system, using mouse and human versions of the protein. IgM is now understood to be shaped like an incomplete hexagon, or a pentagon with a wedge-shaped gap.
"We'll have to revise the textbooks," said Professor Toru Miyazaki, the leader of the laboratory where the IgM structure was identified at the Graduate School of Medicine.
IgM is the first immune system protein that develops in the human fetus and remains the first responder to pathogens throughout life. The structure of IgM was first identified in 1969 as a "five-pointed, star-shaped table" and updated in 2009 to be a five-sided dome or "mushroom cap."
"The original IgM model was made by looking at a few individual molecules by hand with what today we think is a low-resolution microscope. Now we have clearer pictures and the computer can examine thousands of individual IgM molecules," said Miyazaki.
The 2018 discovery of the incomplete hexagon was actually a secondary interest for Miyazaki, originally a medical doctor who has built his research career studying a different protein called apoptosis inhibitor of macrophage (AIM).
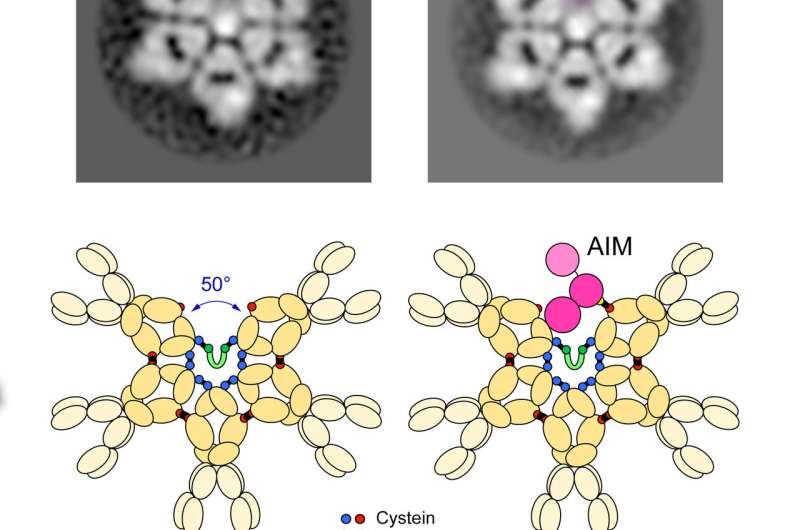
Since identifying the correct shape of IgM, researchers now understand that inactive AIM is nestled inside the gap of the IgM incomplete hexagon. The structural connection between IgM and AIM means that drugs with the ability to regulate the release of AIM could be used to create AIM-based disease therapies.
"We can think of AIM as a fighter jet and IgM as the aircraft carrier ship. When other molecules activate the immune system, IgM releases AIM. The much smaller AIM protein goes around the body to clear away damaged cells and prevent disease," said Miyazaki.
Miyazaki identified AIM in 1999 while working at the Basel Institute for Immunology Institute in Switzerland. Its small size means AIM is easily eliminated from the body by the kidneys and excreted in urine, so staying bound within the larger IgM protects AIM from being removed before it is needed.
AIM is a common molecule in the bloodstream, but it is only active when the body develops a disease. AIM is known to be important for preventing obesity, fatty liver disease, hepatocellular carcinoma (liver cancer), multiple sclerosis (MS), fungus-induced peritonitis (inflammation of the abdominal wall membrane), and acute kidney injury.
The incomplete hexagon structure is still only a 2-D understanding of IgM structure. Miyazaki and his team continue to do additional analysis and hope to report the 3-D structure of IgM soon.
More information: E. Hiramoto, A. Tsutsumi, R. Suzuki, S. Matsuoka, S. Arai, M. Kikkawa, T. Miyazaki, The IgM pentamer is an asymmetric pentagon with an open groove that binds the AIM protein. Science Advances 4: eaau1199 (2018). advances.sciencemag.org/content/4/10/eaau1199
A. FEINSTEIN et al. Conformation of the Free and Antigen-bound IgM Antibody Molecules, Nature (2006). DOI: 10.1038/2241307a0
D. M. Czajkowsky et al. The human IgM pentamer is a mushroom-shaped molecule with a flexural bias, Proceedings of the National Academy of Sciences (2009). DOI: 10.1073/pnas.0903805106
Journal information: Science Advances , Proceedings of the National Academy of Sciences , Nature
Provided by University of Tokyo