Cathode material made from organic molecules enhances the green credentials of rechargeable batteries
Incorporating organic materials into lithium ion batteries could lower their cost and make them more environmentally friendly, A*STAR researchers have found. The team has developed an organic-based battery cathode that has significantly improved electrochemical performance compared to previous organic cathode materials. Crucially, the new material is also robust, remaining stable over thousands of battery charge/discharge cycles.
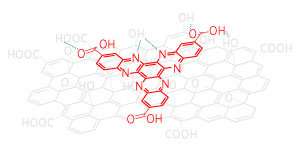
The cathode, the positive electrode in Li-ion batteries, is a critical component. An electron-deficient, rigid organic molecule called hexaazatrinaphthalene (HATN) was previously investigated as an organic cathode material for lithium ion batteries. However, its promising initial performance declined rapidly during use, because the molecule began to dissolve into the battery's liquid electrolyte.
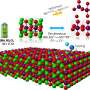
A new cathode material, in which HATN was combined with graphene oxide in a bid to prevent the organic material from dissolving, has now been developed by Yugen Zhang and his colleagues from the A*STAR Institute of Bioengineering and Nanotechnology.
In graphene oxide, a single-atom thick sheet of carbon atoms is partly covered by a layer of oxygen atoms. "Graphene oxide has excellent electronic conductivity, and surface oxygen functionality that may form hydrogen-bonding interactions with HATN," Zhang says. He explains that this made graphene oxide a promising candidate for forming a HATN-graphene oxide nanocomposite.
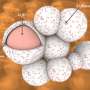
The nanocomposite's performance exceeded expectations. The materials combined to form core-shell nanorods in which the HATN was coated with graphene oxide. "Graphene oxide and HATN formed a very nice composite structure, which solved the dissolution issue of HATN in electrolyte and gave the cathode very good cycling stability," Zhang says. A lithium ion battery using this material as its cathode retained 80 per cent of its capacity after 2000 charge/discharge cycles.
The team saw even better performance when they combined graphene oxide with a HATN derivate called hexaazatrinaphthalene tricarboxylic acid (HATNTA). A battery made from this material retained 86 per cent of its capacity after 2,000 charge/discharge cycles. The improved performance is probably due to the polar carboxylic acid groups on the HATNTA molecule, which attached the molecule even more strongly to the graphene oxide.
The team is continuing to develop new materials to improve the performance of organic cathodes, Zhang says. Aside from investigating alternatives to graphene oxide, the team also is working on HATN-based porous polymers for use as organic cathode materials, which should enhance the flow of ions during battery charge and discharge.
More information: Jinquan Wang et al. Hexaazatriphenylene derivatives/GO composites as organic cathodes for lithium ion batteries, Journal of Materials Chemistry A (2018). DOI: 10.1039/C7TA10232A
Journal information: Journal of Materials Chemistry A