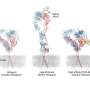
Cryo-electron microscopy characterizes integrins
A collaboration between three labs at UC San Francisco has resulted in an unprecedented look at a member of a vital and ubiquitous class of proteins called integrins (pronounced "INT-uh-grins"). Integrins are associated with fibrosis, scarring and stiffening of tissues that is associated with nearly half of all deaths in developed countries, and yet researchers had no high-resolution structural model of the proteins in their active state. Now, a combination of perseverance, technological achievement and insight has pinned down an elusive moving target.
Older techniques like X-ray crystallography require researchers to undergo laborious processes to pack proteins into crystals before they can make images to determine a protein's structure. This method works best on stationary, rigid, and symmetrical proteins: the opposite of integrins, which are quite flexible in their active form, said Stephen Nishimura, MD, one of the paper's senior authors and a professor of pathology at UCSF.
Integrins are embedded on the surfaces of all animal cells, connecting each cell to its surroundings and allowing it to communicate and respond to external forces. In order to encounter its targets, the new work suggests for the first time that an active integrin bends and sways at a flexible midpoint "like a sunflower seeking the sun," said Nishimura.
To explore an integrin's structure, the team used cryo-electron microscopy, a technique that has recently benefited from major advancements in hardware and software at UCSF. Melody Campbell, Ph.D., worked to visualize one type of integrin protein down to near-atomic precision. She imaged and analyzed the purified and frozen proteins in the lab of Yifan Cheng, Ph.D., a professor of biochemistry and biophysics at UCSF and the other senior author of the study.
But visualizing the protein was only part of the effort. The team's new paper in Nature Structural and Molecular Biology includes Campbell's work, genetic manipulation from Saburo Ito, Ph.D., and protein engineering, purification and expertise from Anthony Cormier, Ph.D. Once the protein was visualized, the researchers validated their structural model by genetically engineering a related integrin that responded to biochemical cues exactly as the team's model predicted, suggesting that their findings extended to many, if not all, integrins.
With antibody engineering pioneers Jim Marks, MD, Ph.D., and Jianlong Lou, Ph.D., both in the Department of Anesthesia at UCSF, the authors have already developed several promising therapeutic antibodies, using the new structure as a template. Some companies are already working with those antibodies to develop treatments for conditions like cancer and fibrosis. But for Nishimura, who has been working with integrins for more than two decades, the detailed model is also personally satisfying: "It's like seeking an old archnemesis, and finally freezing him in his tracks."
More information: Anthony Cormier et al. Cryo-EM structure of the αvβ8 integrin reveals a mechanism for stabilizing integrin extension, Nature Structural & Molecular Biology (2018). DOI: 10.1038/s41594-018-0093-x
Journal information: Nature Structural and Molecular Biology , Nature Structural & Molecular Biology
Provided by University of California, San Francisco