Optimizing lithium-sulfur battery electrolytes for long life
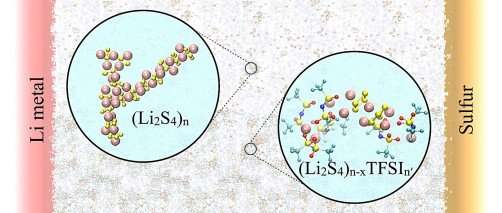
In the future, lithium-sulfur (Li-S) rechargeable batteries could hold more energy and cost less than lithium-ion batteries if they can be made to last longer. A key challenge is preventing the polysulfide ions that must form at the sulfur electrode during discharge from being transported through the liquid electrolyte to the other electrode. Here, researchers discovered how the solubility and transport of polysulfide ions in the liquid electrolyte relates to the chain length of the polysulfide ion, the tendency of the ion to cluster, and the amount of lithium salt present in the electrolyte. It is noteworthy that polysulfides of any chain length or structure dissolved in the liquid electrolyte can contribute to poorer performance of the battery.
Increasing the lifetimes of lithium-sulfur rechargeable batteries requires preventing polysulfide transport between electrodes during operation. Knowledge of how the salt and solvent molecules of the electrolyte interact with polysulfides helps in understanding their solubility. Insights from studies on these interactions could aid in the design of new electrolytes for longer lasting, practical lithium-sulfur rechargeable batteries.
The lithium-sulfur battery has a theoretical energy density much greater than commercial lithium-ion batteries; however, the technology exhibits fade and hence limited battery life. In these batteries, active sulfur species (as dissolved polysulfide ions) migrate to the lithium metal electrode. Then, irreversible reactions involving these polysulfides can occur at the electrode that result in the loss of its ability to store charge. Attempts to protect the metal from the polysulfides have been only partially successful.
Scientists at the Joint Center for Energy Storage Research have focused on tuning the electrolyte composition to control how polysulfides dissolve, move, and interact. Recently, using combined theoretical and experimental studies, the scientists discovered that the solubility of polysulfides (Li2Sx, x=2-8) depend on the chain length of the sulfide ion. Shorter polysulfides form large clusters, with up to fifteen ions in each cluster, which limits their solubility. Longer polysulfides (with more than four sulfur atoms) show higher solubility, but slow transport in the pure solvent. Surprisingly, the addition of a lithium salt to form an electrolyte solution increased the solubility of the short chain polysulfides by providing favorable interactions between the salt and the polysulfides.
The researchers also calculated and measured experimentally the diffusion rates for the lithium ion and other species in the electrolytes at different salt/solvent ratios. High salt concentrations could favorably control how polysulfides dissolve, but resulted in reduced mobility for ionic species and insufficient ionic conductivity. Understanding the structure and dynamics of lithium-sulfur electrolytes provides an opportunity to fine-tune the composition of the electrolyte. With these insights, future electrolytes could be designed to provide efficient transport of ions between the electrodes, while suppressing the dissolution and migration of polysulfide species.
More information: Nav Nidhi Rajput et al. Elucidating the Solvation Structure and Dynamics of Lithium Polysulfides Resulting from Competitive Salt and Solvent Interactions, Chemistry of Materials (2017). DOI: 10.1021/acs.chemmater.7b00068
Journal information: Chemistry of Materials
Provided by US Department of Energy




















