Elucidation of vibration energy of a single molecule in an external force field
The vibration of a molecule on a surface contains critical information on the molecule-surface interaction, crucial for understanding surface phenomena and for important processes like catalysis. It was previously investigated with a scanning probe microscopy, but the probe tip appeared to exert a force on the molecule, affecting the vibration. Here, by combining STM, AFM and model calculations, the experimental results were precisely reproduced; the molecule-surface interactions were weakened by the probe tip proximity.
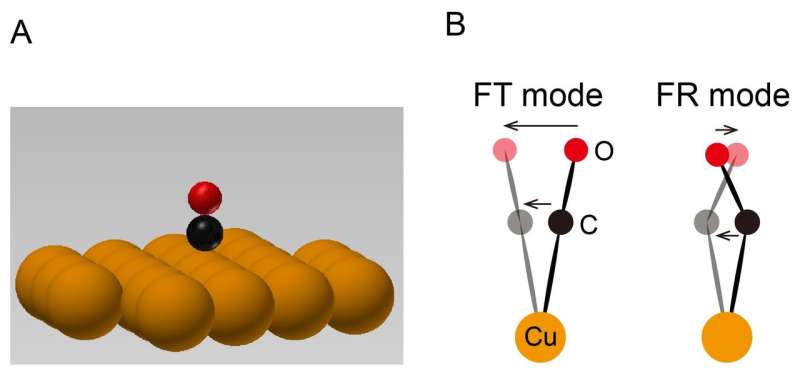
A molecule adsorbed on a surface (Figure 1A) vibrates on the surface (Figure 1B). The vibration energy is determined by the mass of the molecule and by the restoring forces exerted on the molecule. The restoring force originates from the interaction within the molecule and with the surface. By measuring the vibration energy, therefore, we are able to learn details of the interaction of a molecule and a surface. This knowledge is useful in understanding important processes in applied sciences like catalytic reactions that take place on a surface.
Since the vibration energy of a molecule depends to a large extent on the environment of the molecule, it is necessary to measure the vibration energy of an individual molecule to obtain a deep understanding of the interaction of a molecule and a surface, taking into account the environment. For example, a single molecule in isolation on a single crystal surface as shown in Figure 1A is an ideal target of this type of research.
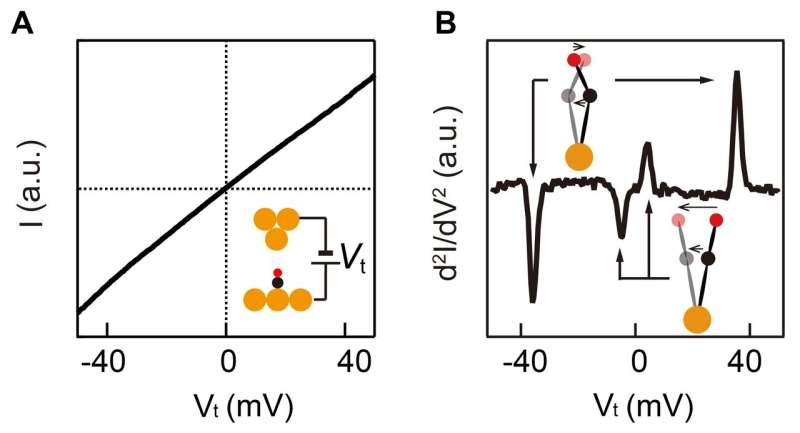
The vibration energy of a single molecule can be investigated, with a scanning tunneling microscope (STM), by placing the metal probe of STM right above the molecule and by precisely measuring the current by changing the voltage applied between the probe of STM and the surface. As shown in Figure 2A, the current (I) and the voltage (V) show an approximately linear relationship, the second derivative of which (V derivative of dI/dV) shows a peak and trough pair as shown in Figure 2B. The peak and trough pair corresponds to the vibration energy of a molecule. Thus, using this method, the vibration energy of a single molecule can be determined.
It was previously reported, however, that when a metal probe was placed very close to a molecule in order to measure the current, the probe tip itself exerted a force on the molecule, affecting its vibration energy. In this study, we have measured the force between the probe and the molecule by atomic force microscope (AFM) and the vibration energy by STM to elucidate their relationship.
The present study was conducted by a collaboration of researchers from Kanazawa University, Japan, University of Regensburg, Germany, and Linnaeus University, Sweden. The experiments were done at University of Regensburg.
The force between a probe and a molecule was measured by using the force sensor developed by Prof. Giessibl, University of Regensburg, Germany, a coauthor of the study. The support that was attached with the force sensor was oscillated at a resonance frequency (about 50 kHz) of the cantilever of the sensor to oscillate the cantilever effectively. A metal probe was attached at the tip of the cantilever, where the probe tip consisted of just one atom. By placing the probe tip in close proximity of a molecule adsorbed on the surface, a force arises between the molecule and the probe tip, which changes the resonance frequency of the cantilever. From such changes, the force between the probe tip and the molecule can be determined. Figure 3A shows the experimental data concerning the force arising between the probe tip and CO molecule adsorbed on a copper surface upon changing the distance between the probe tip and CO molecule; one set of data is compared with another set with a different probe tip. This comparison indicates the difference in the forces exerted on the molecule by the two different probe tips. Each probe tip consists of just one atom, but the difference in the structure behind the single atom affects the forces exerted.
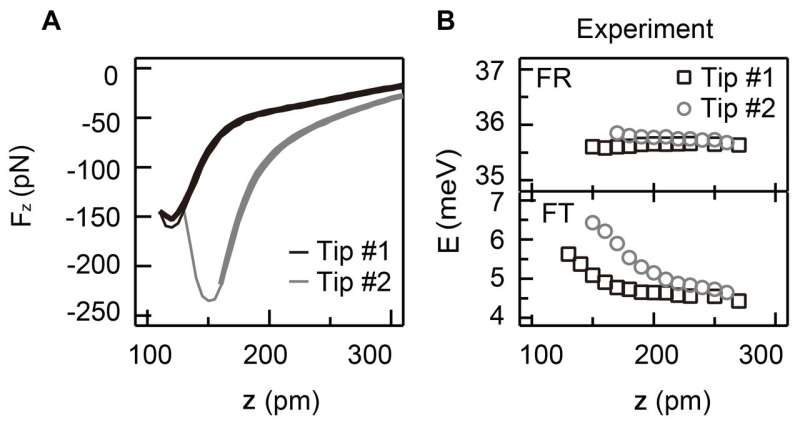
After the force measurements, the vibration energy was investigated by precisely measuring the current that was generated by applying a voltage between the probe tip and the surface. Figure 3B shows the change of the vibration energy upon changing the distance between the probe tip and the molecule. The probe tip that exerts the larger attracting force affects the vibration energy of the molecule to a greater extent.
Next, the experimental results were analyzed using a classical model that considers the vibration of a molecule as a double pendulum. With an ordinary pendulum, gravity provides a restoring force, while in this study, the bonds within the molecule and between the molecule and the surface provided a restoring force. The vibration energy was calculated using this pendulum model with forces arising between the probe tip and the molecule taken into consideration. In addition, it was also taken into consideration that the forces exerted by the probe tip weakened the bonds within the molecule and between the molecule and the surface. This model successfully and precisely reproduced the experimental results.
The present study significantly deepens our understanding of the interaction of a molecule and a surface and of the interaction of a probe tip and a molecule. In this study, a simple molecule, CO, having a very simple molecular structure has been employed as a target of research. It is expected that this study will stimulate further investigation of molecules of more complicated structure and technological importance. It is also expected that the bond between a molecule and a surface would be severed by a metal probe tip, which may be applied to processes inducing chemical reactions.
More information: Norio Okabayashi et al, Vibrations of a molecule in an external force field, Proceedings of the National Academy of Sciences (2018). DOI: 10.1073/pnas.1721498115
Journal information: Proceedings of the National Academy of Sciences
Provided by Kanazawa University