Pattern formation—the paradoxical role of turbulence

The formation of self-organizing molecular patterns in cells is a critical component of many biological processes. Researchers from Ludwig-Maximilians-Universitaet (LMU) in Munich have proposed a new theory to explain how such patterns emerge in complex natural systems.
Many biological processes are crucially dependent on the formation of ordered distributions of specific molecules within cells. These patterns are self-organizing structures that evolve in a predictable fashion in time and space. Perhaps the best known example of intracellular protein patterning is the molecular machinery that orchestrates the regular segregation of complete chromosome sets to the two daughter cells during cell division.
The classical theory of pattern formation is based on chemical systems that are close to equilibrium. But such states are seldom encountered in physical, chemical or biological systems in which self-organized patterning is typically observed. As a rule, these systems are found to be very far from equilibrium, a state maintained by the input of energy. The mechanisms that create and stabilize ordered structures under these conditions are poorly understood. LMU physicists Erwin Frey and Jacob Halatek have now introduced a new theoretical framework that can account for pattern formation in non-equilibrium systems. The new theory is described in the journal Nature Physics.
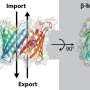
Frey and Halatek focused their attention on dynamical systems that are driven by mass-conserving interactions—i.e., chemical reactions. In biological systems, pattern formation primarily manifests itself in the dynamical redistribution of specific proteins. In many of these systems, the dynamics depends on alterations in the conformations of the protein molecules that enable them to switch between a membrane-bound state and a freely diffusing state in the cell's soluble phase. "What we observe as a protein pattern is usually a specific spatial arrangement, a non-uniform density, of a protein, on a membrane surface," says Halatek.
Pattern formation results from the fact that the distribution of a given protein between the membrane and the cytosolic phase is constantly changing, although its total concentration in the cell remains constant. "The dynamics of pattern formation in such a complex and extended system as a biological cell is, however, very difficult to capture, even in simulations," says Halatek. "That's why we divided the data used in our simulations of pattern formation in large systems into a lattice of much smaller compartments, which are coupled to each other."
The local density of membrane-bound and cytosolic proteins determines the chemical equilibrium in each compartment—such that changes in the ratio of cytosolic to membrane-bound forms of proteins result in a shift of the equilibrium. Halatek and Frey showed that pattern formation is a consequence of these shifts in local chemical equilibria. "The redistribution of the proteins is driven by diffusion. Diffusion on its own would eventually lead to a homogeneous distribution of all protein species throughout the cell volume," says Halatek. It is therefore essential for pattern formation that a diffusion gradient be maintained in the system, so that redistributions of the proteins are always possible. For this reason, pattern formation in biological systems depends on enzymatic reactions that alter the conformations of the proteins concerned, to enable them to bind to the membrane, for instance."
The two physicists applied their new theory to the Min system—a set of three proteins found in the rod-shaped bacterium Escherichia coli, which interact to generate a self-organizing pattern that determines the plane of cleavage during cell division. They observed another consequence of the dynamic destabilization of local equilibria due to mass transport—the emergence of chemical turbulence. "These turbulences, however, do not result in the complete loss of order that classical theories suggest," says Frey. "In our conceptual framework, precisely the opposite occurs. When we destabilize the system, we observe that turbulence develops relatively rapidly. But upon further perturbation, the system undergoes a transition in which it is far from equilibrium, but nevertheless clearly ordered and non-turbulent." Frey and Halatek compare this type of behavior to the effect of a cardiac pacemaker, which counteracts arrhythmias by applying electrical impulses to restore the normal pattern of impulse conduction. "Our model explains how 'pacemakers' can emerge by self-organization in non-equilibrium systems," says Halatek. "In other words, we can provide a clear answer to the question: What part of the 'self' is responsible for the 'organization?' That role is performed by the unstable modes ('control modes') which alter the position and stability of the local equilibria that drive the time-evolution of the system."
More information: J. Halatek et al, Rethinking pattern formation in reaction–diffusion systems, Nature Physics (2018). DOI: 10.1038/s41567-017-0040-5
Journal information: Nature Physics
Provided by Ludwig Maximilian University of Munich