Toxic genetic material's origins discovered
Colibactin, a toxic agent produced by gut bacteria, including certain strains of E. Coli, and thought to contribute to colon cancer, is assembled via a chemical transformation never seen before, according to Northwestern Medicine research published in Nature Chemical Biology.
Prior to these findings, many of the mechanisms behind the molecular assembly of colibactin were largely unknown. The newly discovered process involves a surprising role for a chemical called an enzyme cofactor.
"Normally, cofactors help enzymes achieve catalysis, but they don't usually get this involved," said study co-author Neil Kelleher, PhD, professor of Medicine in the Division of Hematology and Oncology.
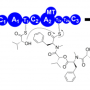
This cofactor, called S-adenosylmethionine (SAM), usually donates a methyl group to an enzyme. In this case, colibactin took that methyl group plus additional atoms to use as building blocks in assembling itself.
"This is a startlingly new enzymatic transformation in nature," Kelleher said. "It's how evolution created this genotoxin, and we're just now figuring out the real details for how this funny little three-membered ring is created."
Learning more about how colibactin forms is key to developing interventions, Kelleher said.
"The more you know about the underlying mechanisms, the more specific your strategy can be," he said.
Sequencing Fungal Genomes for Drug Discovery
Kelleher's genomic investigations have also resulted in the development of a novel platform for analyzing the genomes of fungi, which has the potential to yield scores of new bioactive molecules that could someday be developed into new drugs.
Previously published in Nature Chemical Biology, the multi-site study of the platform, called fungal artificial chromosomes and metabolic scoring (FAC-MS), showed that the approach has the potential to reinvigorate natural drug discovery.
FAC technology breaks up the fungal genome into megabase-sized DNA segments, which contain entire biosynthetic gene clusters that are associated with specialized metabolites. These metabolites represent an enormous untapped reservoir of potential drugs, according to the study. Because FAC-MS analyzes the genome, scientists not only get the metabolites but the genes that created them, giving scientists the ability to scale or alter the new compounds.
"It's a hibernating area that's ready to explode," said Kelleher, also the director of Northwestern Proteomics and a professor of Biochemistry and Molecular Genetics.
More information: Li Zha et al. Colibactin assembly line enzymes use S-adenosylmethionine to build a cyclopropane ring, Nature Chemical Biology (2017). DOI: 10.1038/nchembio.2448
Journal information: Nature Chemical Biology
Provided by Northwestern University