Designing selective reactions to modulate biological processes

Unlike chemical reactions carried out in flasks in the laboratory, which allow researchers to convert reagents into products, usually in organic solvents, in biological environments, everything is much more unpredictable and unstable. Thus, reactions within a living organism proceed in a markedly hostile media: dense, complex, and surrounded by many other adjacent substances that threaten its feasibility (such as amino acids or sugars).
Fortunately, nature has evolved sufficiently to allow these reactions to take place with astonishing selectivity, without interference from any other biological events. These transformations, so fundamental to the cell and to life, are known as bioorthogonal, and are usually promoted by enzymes.
So far, very few non-natural reactions of this type have been identified, but their potential for modifying biological functions in a controlled manner has raised increasing interest among scientists. Thus, chemists have spent years working on the design of novel bioorthogonal reactions which should be compatible with the complexity of biological media.
Now, a team of CiQUS led by Professor José Luis Mascareñas and Dr. Fernando López has managed to develop a new bioorthogonal transformation that allows a selective coupling of two designed molecular fragments, without the interference of any of the molecules that abound in cells and tissues, such as proteins or nucleic acids.
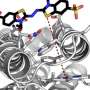
The work published in the prestigious chemistry journal Angewandte Chemie , describes a novel reaction that is programmed to occur only when a ruthenium complex is present in the medium. "Ruthenium acts as a catalyst, it functions as an 'artificial enzyme,'" explains Paolo Destito, the first author of the paper. According to this predoctoral researcher, "with this new method you can select exclusively, within a complex biological media, the two fragments that are intended to be fused, joining them together to generate the product of the desired chemical reaction."
The relevance of this work-still in a preliminary phase, can be estimated by its multiple potential applications. Indeed, Helio Faustino, a former CiQUS researcher, also responsible for the article, says "we are confident that this discovery might eventually provide a very powerful chemical tool to investigate the functioning of cells at a molecular level." José Couceiro, a postdoctoral researcher at the center, points out its importance highlighting that "it could be eventually used to selectively produce bioactive substances or drugs, just where their actions are specifically needed."
However, Professor Mascareñas prefers to be cautious: "We still have a lot to do, we are still working to optimize the reaction and improve its efficiency before demonstrating its effectiveness inside living systems, but there is no doubt that the results are very promising," he says.
Journal information: Angewandte Chemie
Provided by CORDIS