Researchers observe individual cellular receptors at work
Using a revolutionary live-cell microscopy technique, an international team of scientist has for the first time observed individual receptors for hormones and drugs working in intact cells.
G protein-coupled receptors (GPCRs) are among the hottest targets for therapies for conditions such as hypertension, asthma or Parkinson's. These receptors are the sites of action of many hormones and neurotransmitters and allow them to regulate the activity of our cells. As a result, around half of all currently prescribed drugs target these receptors to treat common diseases.
As their name suggests, these receptors have to find and interact with so-called G proteins on the cell membrane in order to initiate and regulate intracellular processes. How and where this happens is not well understood. An international team of scientists from the Universities of Würzburg, Birmingham and Wroclaw has now directly observed and investigated the initial contacts between individual receptors and G proteins and the subsequent events at the surfaces of living cells.
"We were able to show that receptors and G proteins preferentially meet at special sites on the cell membrane, which we call hot spots," says Professor Davide Calebiro, the senior author of the study, which has just appeared in the journal Nature.
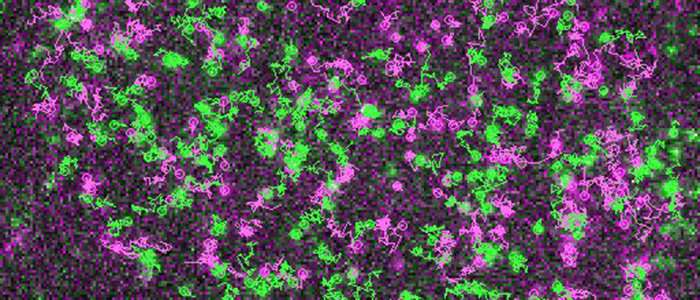
These detailed observations of living cell membranes were made possible by the use of a highly sophisticated method based on single-molecule microscopy. The authors also observed that receptors and G proteins usually remain associated for only a short time. Indeed, most interactions terminated after just one second. Furthermore, the researchers found that the cytoskeleton underneath the cell membrane plays an important role in the formation of the hot spots. The existence of these hot spots for receptor signaling was hitherto unknown.
Davide Calebiro and his co-authors are convinced that the hot spots exert an important influence on receptor signaling by increasing the speed and efficiency of G protein activation. At the same time, they allow the resulting signals to stay local. According to the scientists, their new findings show that "apparently simple biological processes can be highly sophisticated once observed from a short distance." They anticipate that exceptional advances in imaging techniques will lead to a much deeper understanding of these processes in the near future.
According to the authors, these insights provide the opportunity for new therapeutic approaches. "Currently used drugs can only either activate or block the receptors," says Davide Calebiro. In the future, it might be possible to influence these processes much more precisely—for example, by manipulating the mobility of receptors and G proteins on the cell membrane or their interactions at the hot spots.
More information: Titiwat Sungkaworn et al, Single-molecule imaging reveals receptor–G protein interactions at cell surface hot spots, Nature (2017). DOI: 10.1038/nature24264
Journal information: Nature
Provided by University of Würzburg