Direct conversion of rusty stainless steel mesh into stable, low-cost electrodes for potassium-ion batteries
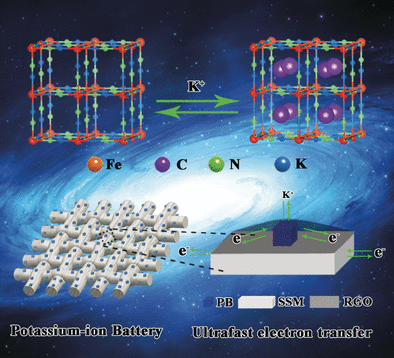
Chinese scientists have made good use of waste while finding an innovative solution to a technical problem by transforming rusty stainless steel mesh into electrodes with outstanding electrochemical properties that make them ideal for potassium-ion batteries. As reported in the journal Angewandte Chemie, the rust is converted directly into a compact layer with a grid structure that can store potassium ions. A coating of reduced graphite oxide increases the conductivity and stability during charge/discharge cycles.
Increasing use of renewable energy requires effective energy storage within the grid. Lithium ion batteries, widely used in portable electronics, are promising candidates. Lithium ion batteries are based on the displacement of lithium ions. While charging, the ions move toward the graphite electrode, where they are stored between the layers of carbon. When discharging, they are released. However, lithium is expensive and reserves are limited. Sodium ion batteries have been explored as an alternative.
"Potassium ions are just as inexpensive and readily available as sodium, and potassium ion batteries would be superior from the electric aspect," reports Xin-Bo Zhang. "However, the significantly larger radius of the potassium ions has posed a problem. Repeated storage and release of these ions destabilizes the materials currently used in electrodes."
Zhang and a team from the Chinese Academy of sciences and Jilin University (Changchun, China) have now found an elegant solution in their use of a waste material to make novel electrodes: rejected stainless steel mesh from filters and sieves. Despite the excellent durability of these grids, harsh conditions do lead to some corrosion. The metal can be reclaimed in a furnace, but this process requires a great deal of money, time, and energy, as well as producing emissions. Says Zhang: "Conversion into electrodes could develop into a more ecologically and economically sensible form of recycling."
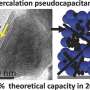
The corroded mesh is dipped into a solution of potassium ferrocyanide (yellow prussiate of potash, known as a fining agent for wine). This dissolves iron, chromium, and nickel ions out of the rust layer. These combine with ferricyanide ions into the complex salt known as Prussian blue, a dark blue pigment that is deposited onto the surface of the mesh as scaffold-like nanocubes. Potassium ions can easily and rapidly be stored in and released from these structures.
The researchers then use a dip-coating process to deposit a layer of graphene oxide (oxidized graphite layers). This layer nestles tightly onto the nanocubes. Subsequent reduction converts the graphene oxide to reduced graphene oxide (RGO), which consists of layers of graphite with isolated oxygen atoms. Zhang explains, "the RGO coating inhibits clumping and detachment of the active material. At the same time, it significantly increases the conductivity and opens ultrafast electron-transport pathways."
In tests, coin cells made with these new electrodes demonstrate excellent capacity, discharge voltages, rate capability, and outstanding cycle stability. Because the inexpensive, binder-free electrodes are very flexible, they are highly suitable for use in flexible electronic devices.
More information: Yun-hai Zhu et al. Transformation of Rusty Stainless-Steel Meshes into Stable, Low-Cost, and Binder-Free Cathodes for High-Performance Potassium-Ion Batteries, Angewandte Chemie International Edition (2017). DOI: 10.1002/anie.201702711
Journal information: Angewandte Chemie , Angewandte Chemie International Edition
Provided by Wiley