Nanoparticles can travel from lungs to blood, possibly explaining risks to heart
Tiny particles in air pollution have been associated with cardiovascular disease, which can lead to premature death. But how particles inhaled into the lungs can affect blood vessels and the heart has remained a mystery. Now, scientists have found evidence in human and animal studies that inhaled nanoparticles can travel from the lungs into the bloodstream, potentially explaining the link between air pollution and cardiovascular disease. Their results appear in the journal ACS Nano.
The World Health Organization estimates that in 2012, about 72 percent of premature deaths related to outdoor air pollution were due to ischemic heart disease and strokes. Pulmonary disease, respiratory infections and lung cancer were linked to the other 28 percent. Many scientists have suspected that fine particles travel from the lungs into the bloodstream, but evidence supporting this assumption in humans has been challenging to collect. So Mark Miller and colleagues at the University of Edinburgh in the United Kingdom and the National Institute for Public Health and the Environment in the Netherlands used a selection of specialized techniques to track the fate of inhaled gold nanoparticles.
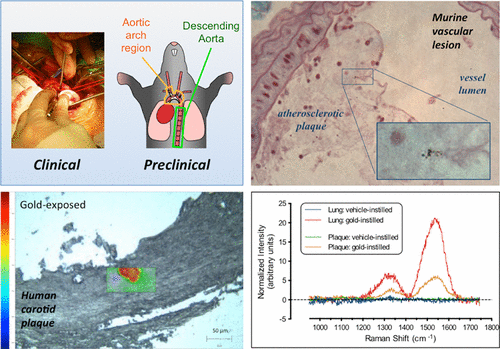
In the new study, 14 healthy volunteers, 12 surgical patients and several mouse models inhaled gold nanoparticles, which have been safely used in medical imaging and drug delivery. Soon after exposure, the nanoparticles were detected in blood and urine. Importantly, the nanoparticles appeared to preferentially accumulate at inflamed vascular sites, including carotid plaques in patients at risk of a stroke. The findings suggest that nanoparticles can travel from the lungs into the bloodstream and reach susceptible areas of the cardiovascular system where they could possibly increase the likelihood of a heart attack or stroke, the researchers say.
More information: Inhaled Nanoparticles Accumulate at Sites of Vascular Disease, ACS Nano, Article ASAP, pubs.acs.org/doi/abs/10.1021/acsnano.6b08551
Abstract
The development of engineered nanomaterials is growing exponentially, despite concerns over their potential similarities to environmental nanoparticles that are associated with significant cardiorespiratory morbidity and mortality. The mechanisms through which inhalation of nanoparticles could trigger acute cardiovascular events are emerging, but a fundamental unanswered question remains: Do inhaled nanoparticles translocate from the lung in man and directly contribute to the pathogenesis of cardiovascular disease? In complementary clinical and experimental studies, we used gold nanoparticles to evaluate particle translocation, permitting detection by high-resolution inductively coupled mass spectrometry and Raman microscopy. Healthy volunteers were exposed to nanoparticles by acute inhalation, followed by repeated sampling of blood and urine. Gold was detected in the blood and urine within 15 min to 24 h after exposure, and was still present 3 months after exposure. Levels were greater following inhalation of 5 nm (primary diameter) particles compared to 30 nm particles. Studies in mice demonstrated the accumulation in the blood and liver following pulmonary exposure to a broader size range of gold nanoparticles (2–200 nm primary diameter), with translocation markedly greater for particles <10 nm diameter. Gold nanoparticles preferentially accumulated in inflammation-rich vascular lesions of fat-fed apolipoproteinE-deficient mice. Furthermore, following inhalation, gold particles could be detected in surgical specimens of carotid artery disease from patients at risk of stroke. Translocation of inhaled nanoparticles into the systemic circulation and accumulation at sites of vascular inflammation provides a direct mechanism that can explain the link between environmental nanoparticles and cardiovascular disease and has major implications for risk management in the use of engineered nanomaterials.
Journal information: ACS Nano
Provided by American Chemical Society


















