Tiny gold particles could be the key to developing a treatment for pancreatic cancer
A diagnosis of pancreatic cancer is often a death sentence because chemotherapy and radiation have little impact on the disease. In the U.S. this year, some 53,000 new cases will be diagnosed, and 42,000 patients will die of the disease, according to the National Institutes of Health. But research now being reported in ACS Nano could eventually lead to a new type of treatment based on gold nanoparticles.
Scientists have previously studied these tiny gold particles as a vehicle to carry chemotherapy drug molecules into tumors or as a target to enhance the impact of radiation on tumors. In addition, Priyabrata Mukherjee and colleagues previously found that gold nanoparticles themselves could limit tumor growth and metastasis in a model of ovarian cancer in mice.
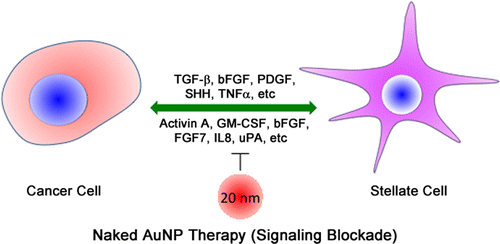
Now, the team has determined that the same holds true for mouse models of pancreatic cancer. But interestingly, the new work revealed details about cellular communication in the area surrounding pancreatic tumors. By interrupting this communication—which is partly responsible for this cancer's lethal nature—the particles reduced the cell proliferation and migration that ordinarily occurs near these tumors. Gold nanoparticles of the size used in the new study are not toxic to normal cells, the researchers note.
More information: Gold Nanoparticle Reprograms Pancreatic Tumor Microenvironment and Inhibits Tumor Growth, ACS Nano, Article ASAP. DOI: 10.1021/acsnano.6b02231
Abstract
Altered tumor microenvironment (TME) arising from a bidirectional crosstalk between the pancreatic cancer cells (PCCs) and the pancreatic stellate cells (PSCs) is implicated in the dismal prognosis in pancreatic ductal adenocarcinoma (PDAC), yet effective strategies to disrupt the crosstalk is lacking. Here, we demonstrate that gold nanoparticles (AuNPs) inhibit proliferation and migration of both PCCs and PSCs by disrupting the bidirectional communication via alteration of the cell secretome. Analyzing the key proteins identified from a functional network of AuNP-altered secretome in PCCs and PSCs, we demonstrate that AuNPs impair secretions of major hub node proteins in both cell types and transform activated PSCs toward a lipid-rich quiescent phenotype. By reducing activation of PSCs, AuNPs inhibit matrix deposition, enhance angiogenesis, and inhibit tumor growth in an orthotopic co-implantation model in vivo. Auto- and heteroregulations of secretory growth factors/cytokines are disrupted by AuNPs resulting in reprogramming of the TME. By utilizing a kinase dead mutant of IRE1-α, we demonstrate that AuNPs alter the cellular secretome through the ER-stress-regulated IRE1-dependent decay pathway (RIDD) and identify endostatin and matrix metalloproteinase 9 as putative RIDD targets. Thus, AuNPs could potentially be utilized as a tool to effectively interrogate bidirectional communications in the tumor microenvironment, reprogram it, and inhibit tumor growth by its therapeutic function.
Journal information: ACS Nano
Provided by American Chemical Society