New method gives accurate picture of gas storage by microscopic cages

A computational method to quantify the adsorption of gas by porous zeolites should help labs know what to expect before they embark upon slow, costly experiments, according to researchers at Rice University.
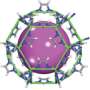
The new method created by engineers in Rice's Multiscale Materials Modeling Lab accurately calculated the ability of two zeolites, small cage-like molecules with enormous surface area, to trap and store gas molecules.
Among other possibilities, the work could help in the race to meet Department of Energy (DOE) standards that call for the creation by 2015 of materials that can hold 5.5 percent of their weight in hydrogen to fuel vehicles.
"We think we can get there," said Rice materials scientist Rouzbeh Shahsavari, who calculated capacities for two of what he called "remarkably large and colossal cages" and found that one comes close to the mark.
The study by Shahsavari, graduate student Navid Sakhavand and former Rice postdoctoral researcher Prakash Muthuramalingam, now a postdoctoral researcher at Université Paris-Est, appears online in the American Chemical Society's Journal of Physical Chemistry.
The lab analyzed a dizzying array of potential interactions for two synthetic microporous materials known as zeolitic imidazolate frameworks, ZIF-95 and ZIF-100. Those "colossal cages" may be only nanometers wide, but the molecules they can store that the lab looked at—hydrogen, methane and nitrogen – are much smaller. The zeolites' enormous surface area inside and out gives gas molecules plenty of room to bind.
Aside from storing hydrogen for fuel, ZIFs show potential for size-selective catalysis, environmental remediation and for use as molecular sieves. "Imagine people are designing fit-for-purpose ZIFs," Sakhavand said. "Before jumping into the experiment and synthesizing them, we can help them rapidly screen the gas uptake for each particular ZIF at various temperatures and pressures."
The researchers' primary goal was to prove the accuracy of their method when compared with a host of experimental results on hydrogen storage carried out elsewhere. Shahsavari said the researchers modeled the interactions between molecules of the three gases with each other and with the binding ligands in the zeolites at 77 and 300 kelvins (-321 and 80 degrees Fahrenheit, respectively) and at various pressures.
For hydrogen, they determined that both zeolites stored about three times as much gas at 77 K and at 100-bar pressure (100 times that of the atmosphere at sea level) than they would at room temperature. ZIF-100, in particular, adsorbed 3.4 percent of its weight in hydrogen, which approaches the DOE standard, Shahsavari said.
"We didn't reach that DOE target with this design, but if we can functionalize the ZIFs by adding ligand-binding moieties (the functional groups in a molecule)into the pore space, then we might be able to. We're working on that," he said.
They were also able to calculate both subtle and significant differences between the adsorptive qualities based on various input parameters of gas, pressure, temperature and type of zeolite. For example, they came to the counterintuitive conclusion that ZIF-100, the larger of the two zeolites, could adsorb more small-molecule hydrogen but fewer of the larger methane molecules than ZIF-95 under similar conditions.
"So our method not only accurately predicts the properties of these porous materials, but also provides fundamental insights that can be leveraged to further improve their properties," Shahsavari said.
The Rice lab's method involved several steps. First, the team performed first-principle calculations to describe the very weak atomic interactions – the van der Waals-related London dispersion forces—among each of the three types of gas molecules and the two ZIFs. The next step used those results to align the potentials among various atomic pairs. Those were plugged into large-scale Monte Carlo simulations to predict how much of each gas each porous zeolite could adsorb.
"Because we combined two methods, each appropriate for a different length scale, we were able to predict the maximum capacity of these materials with high accuracy while maintaining reasonable computational time," Shahsavari said.
The method may seem simple, but calculating integrative forces between thousands of gas molecules and each ZIF was not. It took the combined power of Rice's DAVinCI and SUGAR supercomputers to find results for all the variations. Even so, calculations for a single data point – one molecule, one zeolite, one temperature – often took 96 processing cores three days to complete.
Shahsavari said the method should also be good for analyzing the potential for zeolites as membranes to separate gases. "It can work not only for single molecules, but also gas mixtures," he said. "This provides a good computational framework so one can do rapid screening for the desired properties."
More information: pubs.acs.org/doi/abs/10.1021/jp408075y
Journal information: Journal of Physical Chemistry A
Provided by Rice University












.jpg)







