The observation of truly metallic behavior in an organic material heralds a new generation of electronic devices
When we think about metals, objects like copper wires and sheets of iron spring to mind. However, organic materials—those based, as all living matter, on carbon and oxygen atoms—can also exhibit metallic behavior. Some organic compounds have been established as good electric conductors, but these systems can be full-fledged metals as Reizo Kato of the RIKEN Advanced Science Institute, Wako, and co-workers in Japan and China have shown. They found unambiguous signatures in an organic compound which establish that the material behaves at low temperatures precisely like most metals.
The interest in 'organic metals' is fuelled by the prospect of technological applications reaching from stretchable electronics to bio-integrated devices. However, almost 40 years after the first discovery of organic metals, a number of fundamental aspects remain to be explored in these materials. In particular, until now it has never been shown that organic metals behave according to the so-called Fermi-liquid theory—the model that describes the behavior of most metals at low temperatures.
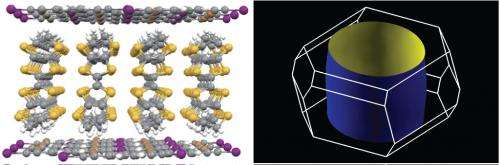
Kato and his colleagues have filled this gap. The team observed the signatures of a Fermi liquid in a compound known as (BEDT-TTF)3Br(pBIB) (Fig. 1, left). "In general, organic conductors are fragile and vulnerable to light irradiation," says Kato. "But over a period of more then ten years our team has made methodological advances—in particular in the area of photoelectron spectroscopy—that allowed us to reduce the disruptive factors." The team's know-how enabled them to successfully conduct a series of experiments in which they showed that at low temperatures the electrons in (BEDT-TTF)3Br(pBIB) indeed behave in the same characteristic manner as they do in a conventional metal (Fig. 1, right).
These findings call for revisiting a number of earlier experiments that indicated that the electrons in organic materials behave differently from a Fermi liquid. But most importantly, the work of Kato and his colleagues provides a sound foundation for understanding organic metals, which in turn should pave the way toward practical applications. "This project will provide important information for understanding electronic processes and designing organic materials," says Kato. Among the organic metals, the (BEDT-TTF)3Br(pBIB) system and related compounds are particularly interesting as they are characterized by an architecture in which two-dimensional conducting layers are separated by insulating supramolecular networks. Such network structures may serve as the building blocks for functional molecular materials, including computing and memory elements for electronic devices.
More information: Kiss, T., et al. Quasiparticles and Fermi liquid behaviour in an organic metal. Nature Communications 3, 1089 (2012). www.nature.com/ncomms/journal/ … full/ncomms2079.html
Journal information: Nature Communications
Provided by RIKEN