Researchers unravel structure of 'Sputnik' virus, could have implications for treatment
(Phys.org)—An international team of researchers has determined key structural features of the Sputnik virus, thought to play a role in illnesses caused by eating raw fish.
Findings also may help scientists learn details about the infection mechanisms in other viruses.
Sputnik is referred to as a "satellite virus" because it can only be propagated in combination with another virus, in this case the mimivirus, which infects amoebas. While inside the single-cell amoeba, the DNA for both Sputnik and mimivirus are replicated, allowing the viruses to multiply.
The amoeba host implies that both mimivirus and Sputnik are frequently present in aquatic food, causing disease when humans eat raw fish.
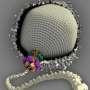
The scientists used a powerful electron microscope to determine the basic design of the Sputnik virus's outer shell, or capsid, and also of the hundreds of smaller units called capsomers making up this shell.
"To be able to use an electron microscope to see near atomic-scale detail of a virus is a major achievement," said Michael Rossmann, Purdue University's Hanley Distinguished Professor of Biological Sciences. "It's an important technical milestone."
The findings are detailed in a research paper that appeared online in October in the journal Proceedings of the National Academy of Sciences. The paper's lead author was Purdue postdoctoral researcher Xinzheng Zhang.
Sputnik, like many other viruses, has a capsid that has icosahedral symmetry, a roughly spherical shape containing 20 triangular faces. This capsid is assembled from 1,620 nearly identical protein subunits organized into 260 hexagonal capsomers and 12 pentagonal capsomers. Each protein subunit is folded into a "double jelly-roll" configuration found in many virus capsids.
Sputnik attaches to and enters host amoebas, encountering a more acidic environment that apparently induces changes in the structure of the virus's protein capsid, releasing the virus's genome, Rossmann said. The research results suggest a mechanism of Sputnik DNA ejection that probably also occurs in other large icosahedral double jelly-roll viruses such as adenoviruses, which cause upper respiratory infections.
"The best way to design effective treatments for viral infections is to first learn what the infection mechanisms are," Rossmann said. "So these findings might offer a long-term benefit."
Researchers studied the structure using the Titan electron microscope, housed in Purdue's Wayne T. and Mary T. Hockmeyer Hall of Structural Biology.
"The Titan instrument has allowed us to do some things that previously haven't been possible," he said.
The researchers used a method called cryoelectron microscopy to determine three-dimensional structural details, identifying most of the amino acids making up the capsid proteins.
"We already knew the sequence of amino acids, but now we know the three-dimensional structure of the three-dimensional fold of the complete capsid protein and its organization in the virus," Rossmann said.
Zhang also discovered a flaw in the previously proposed genome sequence of the virus.
"This is important because it tells us more about the nature of the virus," Rossmann said.
Mimivirus is the largest known virus. It has been called a possible "missing link" between viruses and living cells. The virus infects amoeba, but it is thought to possibly be a human pathogen because antibodies to the virus have been discovered in pneumonia patients. However, many details about the virus remain unknown.
More information: 3.5-Å Resolution Structure of the Sputnik Virophage, Xinzheng Zhanga, Siyang Suna, Ye Xianga, Jimson Wonga, Thomas Klosea, Didier Raoultb, and Michael G. Rossmanna, PNAS, 2012.
ABSTRACT
"Sputnik" is a dsDNA virus, referred to as a virophage, that is co-assembled with Mimivirus in the host amoeba. We have used cryo-EM to produce an electron density map of the icosahedral Sputnik virus at 3.5-Å resolution, sufficient to verify the identity of most amino acids in the capsid proteins and to establish the identity of the pentameric protein forming the fivefold vertices. It was also shown that the virus lacks an internal membrane. The capsid is organized into a T = 27 lattice in which there are 260 trimeric capsomers and 12 pentameric capsomers. The trimeric capsomers consist of three double "jelly-roll l" major capsid proteins creating pseudohexameric capsomer symmetry. The pentameric capsomers consist of five single jelly-roll proteins. The release of the genome by displacing one or more of the pentameric capsomers may be the result of a low-pH environment. These results suggest a mechanism of Sputnik DNA ejection that probably also occurs in other big icosahedral double jelly-roll viruses such as Adenovirus. In this study, the near-atomic resolution structure of a virus has been established where crystallization for X-ray crystallography was not feasible.
Journal information: Proceedings of the National Academy of Sciences
Provided by Purdue University