Cell growth protein Ras forms a 'pair' on the cell membrane

German biophysicists at Ruhr-Universitaet-Bochum in collaboration with the MPI Dortmund have for the first time measured the orientation of the Ras protein bound to the cell membrane.
The RUB team combined the use of three biophysical methods - infrared spectroscopy, computer simulations and fluorescence measurements - and came to the surprising conclusion that two Ras molecules form a pair to take an upright position on the membrane. It was previously assumed, based on computer simulations, that the protein is located horizontally on the membrane as single molecule. Ras is the central "switch" for cell growth, and malfunction of this protein is an important factor in the development of cancer.
"These results put a completely new light on the nano-cluster formation of Ras at the membrane," said Professor Dr. Klaus Gerwert from the RUB Faculty of Biology and Biotechnology.
The study was chosen as the cover story for the Biophysical Journal.
Orientation affects protein interactions
The orientation of a protein affects its possible interactions with other proteins. "This is similar to comparing the situations of a guest being welcomed with open arms and the host lying on the couch during the greeting," illustrates Dr. Jörn Güldenhaupt, who conducted the orientation measurements. Few biophysical methods allow the protein orientation to be determined. ATR-FTIR spectroscopy, which has been established by the Chair for Biophysics, is one of them.
Ras molecules are mutually supportive
The false assumption that Ras lies on the membrane was based on earlier computer simulations. Till Rudack from the Bochum research team also took a virtual look at Ras. The result: A single upright Ras molecule very quickly falls over and seems to lie on the membrane. "Something must have supported the Ras in our measurements," said Till Rudack. "And that could only be another Ras molecule that was not present in the simulation." In fact, further computer simulations of two mutually supportive Ras molecules yielded a stable upright orientation – which would fit the experimental results.
Fluorescence resonance energy transfer: a molecular yardstick
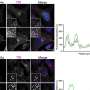
The team confirmed the results with another piece of experimental evidence using "FRET" (fluorescence resonance energy transfer). This is currently the best method for detecting interactions between two proteins. Here, researchers mark the Ras proteins with two different dyes. If the proteins interact, they are very close together so that energy is transferred from the one dye to the other. As with a yardstick, the distance between the proteins can be measured from the ratio of the transferred energy. For the Ras-Ras interaction, the biophysicists determined a distance of 4.6 nanometres, or millionths of a millimetre. This corresponded exactly to the distance they had predicted with their computer simulations for a "double-Ras".
Stronger in the group
Earlier studies had shown that Ras molecules are often concentrated in small groups. These so-called nano-clusters consist of four to ten Ras proteins. Up until now, it was assumed that other proteins have to mediate the formation of clusters. "We were able to demonstrate for the first time that Ras itself is actively involved," said Dr. Carsten Kötting, an Assistant Professor. The clustering is a great advantage for Ras. The proteins are able to pass on a signal more clearly in the group, i.e. with fewer errors. The SOS protein, for example, always transmits one signal simultaneously to two Ras molecules. If Ras is present in a double form (as a dimer), this step is much easier. An understanding of the stereo structure of Ras will allow us to adopt novel approaches in drug development. "So far, no drugs have been discovered which act directly on Ras," said Klaus Gerwert. "Ras is to be considered undruggable. The Ras-Ras interface could be a new starting point, however, in the development of Ras drugs."
More information: J. Güldenhaupt, T. Rudack, P. Bachler, D. Mann, G. Triola, H. Waldmann, C. Kötting, K. Gerwert (2012): N-Ras forms dimers at POPC membranes, Biophysical Journal, doi: 10.1016/j.bpj.2012.08.043
Journal information: Biophysical Journal
Provided by Ruhr-Universitaet-Bochum