Hot attraction in bimetals: A cyano-bridged vanadium-niobium bimetal assembly with a Curie temperature of 210 K
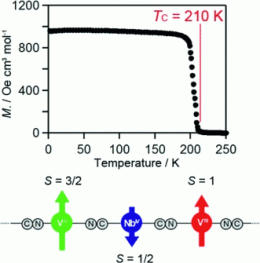
Cyano-bridged bimetal assemblies attract attention because of their magnetic properties such as photomagnetization, humidity-induced magnetization, and nonlinear magneto-optical effect, which make them suitable for many applications. A high Curie temperature is an asset for the use of such magnetic compounds in functional materials. Hexa-, hepta-, and octacyanometalates have been shown to have high Curie temperatures as a result of the high coordination number of their metal centers and the large superexchange interactions due to their diffuse 4d or 5d orbitals. Shin-ichi Ohkoshi and his co-workers at the University of Tokyo report the compound with the highest Curie temperature among octacyano-bridged bimetal assemblies in the Short Communication published in the European Journal of Inorganic Chemistry.
On the basis of initial studies indicating that an increased stoichiometry of vanadium(II) led to a higher Curie temperature in vanadium hexacyanochromate systems, Ohkoshi et al. used a small amount of VIII as catalyst to convert a higher proportion of VII in a similar system. The magnetic properties of the resulting octacyano-bridged vanadium–niobium bimetal assembly were investigated. The compound, whose formula was determined to be K0.59VII1.59VIII0.41[NbIV(CN)8] ·(SO4)0.50·6.9H2O, is ferrimagnetic, and the spins on VII and VIII are antiparallel with respect to the spin on NbIV. Its Curie temperature is 210 K. This high value is a result of the enhanced superexchange interaction through the VII–NC–NbIV pathway.
This study reports a strategy to synthesize magnetic materials with high Curie temperature to enhance the suitability of their magnetic properties for applications.
More information: Shin-ichi Ohkoshi, A Cyano-Bridged Vanadium–Niobium Bimetal Assembly Exhibiting a High Curie Temperature of 210 K, European Journal of Inorganic Chemistry, dx.doi.org/10.1002/ejic.201101219
Journal information: European Journal of Inorganic Chemistry
Provided by Wiley

















