Three-part handoff delivers proteins to membrane surface
The delivery system for an important class of proteins in the cell membrane can be fully replicated with a mere three components, according to a new study.
Tail-anchored proteins, the molecular machines that make up approximately five percent of the membrane proteins in a cell, are known to have their own special pathway for trafficking to the membrane after construction. New research from the University of Chicago and the National Institutes of Health blending structural and functional experiments finds that these proteins can be delivered to the membrane via a simple three-part system.
This deeper understanding of the tail-anchored protein pathway could have significance for the development of new drugs and bioengineering methods. Researchers studying how other types of proteins are delivered to the membrane may also benefit from comparison with this specialized pathway as it is further manipulated and dissected.
"What we are really excited about is the prospect of having a completely defined, completely synthetic controlled system," said Robert Keenan, PhD, Assistant Professor of Biochemistry and Molecular Biology at the University of Chicago. "Now we can really start asking detailed mechanistic questions."
A team of six scientists from the University of Chicago and the National Institute of Child Health and Human Development, led by Keenan and Ramanujan Hegde, MD, Ph.D., published the findings on Wednesday in the journal Nature.
Proteins are put together by ribosomes, which read DNA instructions and link amino acids together into their final form. But many proteins must be delivered from the ribosome to the endoplasmic reticulum (ER), where they are packaged and sent to their final destination.
The majority of membrane proteins navigate this route by using the "co-translational pathway," where the ribosome builds the protein directly into the ER membrane. But tail-anchored (TA) proteins, so named because only a single, small stretch at the "tail" end of the protein sits in the membrane, are known to use a different delivery system.
"TA proteins play all sort of important roles in a variety of different cellular functions," Keenan said. "If you screw this pathway up, bad things will happen. At that level they are just fundamentally important."
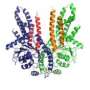
The first component from this new system was identified in 2007 by Hegde (now at the MRC Laboratory of Molecular biology in Cambridge, England). That protein, called Get3 in yeast, was subsequently discovered to interact with two proteins called Get1 and Get2. But researchers hadn't yet determined how these components worked, and whether these three alone could account for TA protein targeting.
Leading the collaboration between Keenan and Hegde's laboratories, co-first authors Agnieszka Mateja of the University of Chicago and Malaiyalam Mariappan of the National Institute of Child Health and Human Development created a synthetic system containing only Get1, Get2, Get3, and a TA protein substrate. The substrate was successfully delivered to the endoplasmic reticulum membrane, confirming that the three-part system was sufficient for trafficking.
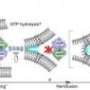
The scientists then deleted or modified specific pieces of the Get proteins to see how these elements work together to move a tail-anchored protein to its proper position in the cell membrane. The new model of the pathway includes both Dr. Octopus-like hooks, a handoff between two closely partnered proteins, and an elegant system for recycling.
1. A complex of two Get3's bound to two molecules of ATP form a "groove" of the right size and chemical properties to capture a tail-anchored protein (the "substrate") in the cytosol.
2. Once the substrate is safely nestled in the groove, "hooks" on the end of Get2 grab the complex, and bring it to the membrane. The long, flexible arms of Get2 allow it to function in a way that Keenan jokingly says is "like Dr. Octopus."
3. Next, Get2 executes a football-style handoff to the adjacent Get1 protein. Binding to Get1 causes the two Get3s to partially "unzip," wedging open the groove and releasing the tail-anchored substrate for insertion into the membrane.
4. Finally, new ATP molecules bind to Get3 causing it to zip back up into the closed form. This releases it from Get1 so that it can initiate another round of protein delivery in the cytosol.
"We have a minimal system, completely purified, that's only three components plus the substrate," Keenan said. "Now we can basically do whatever we want. We can make mutants or chemical modifications, and then we can reconstitute the system and ask, 'does it work?' And if it doesn't work, we can ask where in this process does it actually fail, and why."
Some steps of the pathway remain incomplete, such as how the tail-anchor of the protein is finally inserted into the membrane after it is released by Get3. But with the purified system, researchers can begin exploring these questions, and comparing the TA protein pathway to the more complex co-translational pathway.
As the delivery systems for proteins of all types are better understood, scientists can then use these systems to create better drugs and manipulate cells for bioengineering purposes. For example, some viruses are thought to exploit protein delivery pathways, and understanding the details of trafficking may suggest new ways of defending cells against infection.
"The more we understand about different targeting pathways, the better our ability to successfully target proteins where we want," Keenan said. "Right now, there's no killer app, but you can imagine a lot of potential uses."
More information: The study, "The mechanism of membrane-associated steps in tail-anchored protein insertion," will be published August 24th online by Nature.
Provided by University of Chicago Medical Center