Scientists finely control methane combustion to get different products
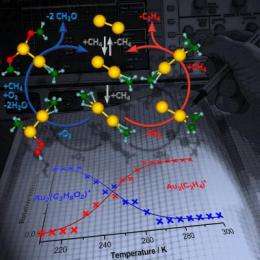
Scientists have discovered a method to control the gas-phase selective catalytic combustion of methane, so finely that if done at room temperature the reaction produces ethylene, while at lower temperatures it yields formaldehyde. The process involves using gold dimer cations as catalysts — that is, positively charged diatomic gold clusters. Being able to catalyze these reactions, at or below room temperature, may lead to significant cost savings in the synthesis of plastics, synthetic fuels and other materials. The research was conducted by scientists at the Georgia Institute of Technology and the University of Ulm. It appears in the April 14, 2011, edition of The Journal of Physical Chemistry C.
"The beauty of this process is that it allows us to selectively control the products of this catalytic system, so that if one wishes to create formaldehyde, and potentially methyl alcohol, one burns methane by tuning its reaction with oxygen to run at lower temperatures, but if it's ethylene one is after, the reaction can be tuned to run at room temperature," said Uzi Landman, Regents' and Institute Professor of Physics and director of the Center for Computational Materials Science at Georgia Tech.
Reporting last year in the journal Angewandte Chemie International Edition, a team that included theorists Landman and Robert Barnett from Georgia Tech and experimentalists Thorsten Bernhardt and Sandra Lang from the University of Ulm, found that by using gold dimer cations as catalysts, they can convert methane into ethylene at room temperature.
This time around, the team has discovered that, by using the same gas-phase gold dimer cation catalyst, methane partially combusts to produce formaldehyde at temperatures below 250 Kelvin or -9 degrees Fahrenheit. What's more, in both the room temperature reaction-producing ethylene, and the formaldehyde generation colder reaction, the gold dimer catalyst is freed at the end of the reaction, thus enabling the catalytic cycle to repeat again and again.
The temperature-tuned catalyzed methane partial combustion process involves activating the methane carbon-to-hydrogen bond to react with molecular oxygen. In the first step of the reaction process, methane and oxygen molecules coadsorb on the gold dimer cation at low temperature. Subsequently, water is released and the remaining oxygen atom binds with the methane molecule to form formaldehyde. If done at higher temperatures, the oxygen molecule comes off the gold catalyst, and the adsorbed methane molecules combine to form ethylene through the elimination of hydrogen molecules.
In both the current work, as well as in the earlier one, Bernhardt's team at Ulm conducted experiments using a radio-frequency trap, which allows temperature-controlled measurement of the reaction products under conditions that simulate realistic catalytic reactor environment. Landman's team at Georgia Tech performed first-principles quantum mechanical simulations, which predicted the mechanisms of the catalyzed reactions and allowed a consistent interpretation of the experimental observations.
In future work, the two research groups plan to explore the use of multi-functional alloy cluster catalysts in low temperature-controlled catalytic generation of synthetic fuels and selective partial combustion reactions.
Provided by Georgia Institute of Technology


















