This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Chemists stabilize ethylene on silver in search for better ethylene purification technology
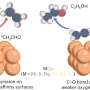
![Ethylene responsive trinuclear silver(I)-pyrazolate [Ag–CF3]3 that undergoes structural changes upon addition of ethylene to form [Ag–CF3(C2H4)]2 and reverts to [Ag–CF3]3 upon removal of ethylene. Credit: Chemical Science (2023). DOI: 10.1039/D3SC04182D Reversible solid-gas reactions of ethylene responsive silver pyrazolates](https://scx1.b-cdn.net/csz/news/800a/2024/reversible-solid-gas-r.jpg)
Production of ethylene is one of the most important chemical processes used today, with about 300 million metric tons of the tiny chemical produced each year. Ethylene gas is used to create everyday items like shopping bags and plastic film packaging.
However, ethylene production consumes massive amounts of energy; by some estimates, methods used to purify gases like ethylene are responsible for about 0.8% of the world's total carbon emissions. Ethylene must be separated from unwanted byproducts through steam cracking, a process that breaks down hydrocarbons by refining petroleum or natural gas.
A team of UTA chemists led by Rasika Dias, professor and chair of chemistry and biochemistry at The University of Texas at Arlington, has found a method that could make these processes more sustainable.
In their recent findings, published in the journal Chemical Science, Dias reports on a type of silver containing material that can absorb ethylene in its solid state, while undergoing remarkable changes in its structure. Such shape-shifting molecules could lead to sustainable ways to catch, purify and release gaseous ethylene.
"My team and I have been hard at work trying to find more sustainable ways to separate, purify and trap ethylene since the chemical is so commercially important to our economy, from the petrochemical industry to agriculture," Dias said.
![Molecular structure of [Ag–CF3]3·CH2Cl2 (top) and [Ag–CF3·(C2H4)]2 (bottom) obtained from solution process and single crystal X-ray diffraction studies. Credit: Chemical Science (2023). DOI: 10.1039/D3SC04182D Chemists stabilize ethylene on silver in search for better ethylene purification technology](https://scx1.b-cdn.net/csz/news/800a/2024/chemists-stabilize-eth.jpg)
The research team included UTA graduate student Devaborniny Parasar and scientist Mukundam Vanga and colleagues from Argonne National Laboratory in Argonne, Illinois; Stony Brook University in Stony Brook, N.Y.; Universidad San Sabastian in Santiago, Chile; and Taras Shevchenko National University in Kyiv, Ukraine.
"The magnitude and speed of structural changes gaseous ethylene propels on silver ion containing solids are quite unbelievable and have not been explored in such intricate detail," Dias said. "It is also challenging to stabilize molecules with ethylene on silver as they create weak bonds with each other. This work also sheds light on our copper-based ethylene purification technology."
In this research, the team used innovative, single crystal X-ray and powder X-ray diffraction techniques to get a clear understanding of the process "live" in molecular form, including seeing the shapes of the molecules with and without ethylene. The results of the experiment were then studied using detailed computational techniques, which led to the insight that silver and ethylene could be successfully stabilized in various forms.
"Our research is exciting because it shows for the first time live ethylene-driven chemistry in solid, crystalline materials," Dias said. "Although our work is preliminary, it has huge implications for how we can work to make raw materials for plastic creation more environmentally friendly."
More information: H. V. Rasika Dias et al, In situ studies of reversible solid–gas reactions of ethylene responsive silver pyrazolates, Chemical Science (2023). DOI: 10.1039/D3SC04182D
Journal information: Chemical Science
Provided by University of Texas at Arlington