This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Engineers boost efficiency for converting greenhouse gas into ethylene
Engineers at the University of Cincinnati have created a more efficient way of converting carbon dioxide into valuable products while simultaneously addressing climate change.
In his chemical engineering lab in UC's College of Engineering and Applied Science, Associate Professor Jingjie Wu and his team found that a modified copper catalyst improves the electrochemical conversion of carbon dioxide into ethylene, the key ingredient in plastic and a myriad of other uses.
Ethylene has been called "the world's most important chemical." It is certainly among the most commonly produced chemicals, used in everything from textiles to antifreeze to vinyl. The chemical industry generated 225 million metric tons of ethylene in 2022.
Wu said the process holds promise for one day producing ethylene through green energy instead of fossil fuels. It has the added benefit of removing carbon from the atmosphere.
"Ethylene is a pivotal platform chemical globally, but the conventional steam-cracking process for its production emits substantial carbon dioxide," Wu said. "By utilizing carbon dioxide as a feedstock rather than depending on fossil fuels, we can effectively recycle carbon dioxide."
The study was published in Nature Chemical Engineering.
Wu's students, including lead author and UC graduate Zhengyuan Li, collaborated with Rice University, Oak Ridge National Laboratory, Brookhaven National Laboratory, Stony Brook University and Arizona State University.
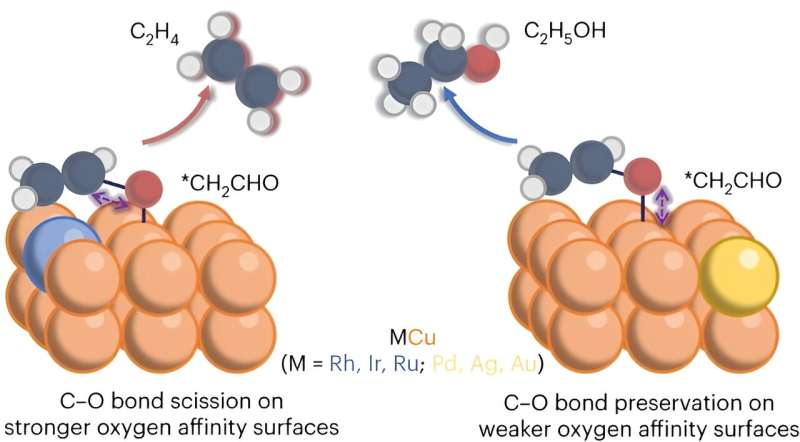
The electrocatalytic conversion of carbon dioxide produces two primary carbon products, ethylene and ethanol. Researchers found that using a modified copper catalyst produced more ethylene.

"Our research offers essential insights into the divergence between ethylene and ethanol during electrochemical CO2 reduction and proposes a viable approach to directing selectivity toward ethylene," lead author Li said.
"This leads to an impressive 50% increase in ethylene selectivity," Wu said. "Ideally, the goal is to produce a single product rather than multiple ones."
Li said the next step is refining the process to make it more commercially viable. The conversion system loses efficiency as byproducts of the reaction such as potassium hydroxide begin forming on the copper catalyst.
"The electrode stability must be improved for commercial deployment. Our next focus is to enhance stability and extend its operation from 1,000 to 100,000 hours," Li said.

Wu said these new technologies will help make the chemical industry greener and more energy efficient.
"The overarching objective is to decarbonize chemical production by utilizing renewable electricity and sustainable feedstock," Wu said. "Electrifying the conversion of carbon dioxide to ethylene marks a significant stride in decarbonizing the chemical sector."
More information: Zhengyuan Li et al, Directing CO2 electroreduction pathways for selective C2 product formation using single-site doped copper catalysts, Nature Chemical Engineering (2024). DOI: 10.1038/s44286-023-00018-w
Journal information: Nature Chemical Engineering
Provided by University of Cincinnati