This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Scientists discover that the natural purification of groundwater is enhanced by nitrate
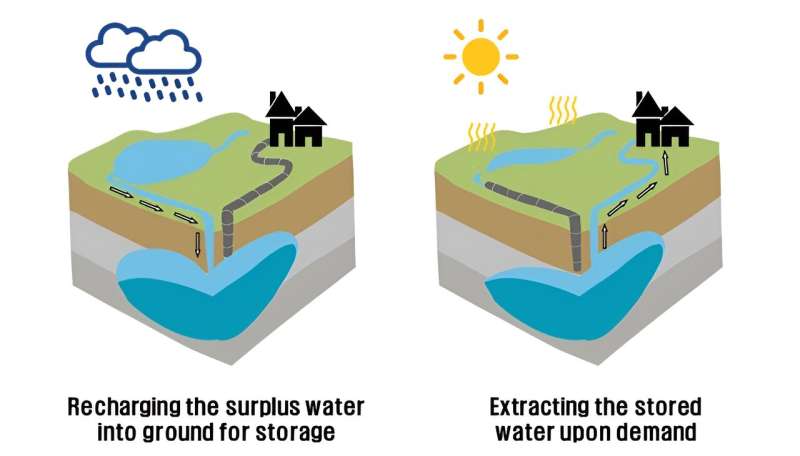
In recent years, the world has been experiencing floods and droughts as extreme rainfall events have become more frequent due to climate change. For this reason, securing stable water resources throughout the year has become a national responsibility called "water security," and "Aquifer Storage Recovery (ASR)", which stores water in the form of groundwater in the ground when water resources are available and withdraws it when needed, is attracting attention as an effective water resource management technique.
A team of researchers including Dr. Seunghak Lee, Jaeshik Chung, and Sang Hyun Kim from the Water Resources Cycle Research Center has discovered that the natural purification of groundwater is enhanced by nitrate, a known pollutant.
In order to apply ASR techniques in practice, it is very important to predict and manage the quality of recharged water, and this research is expected to mark a turning point in the water quality management strategy of ASR systems. The study is published in the journal Water Research.
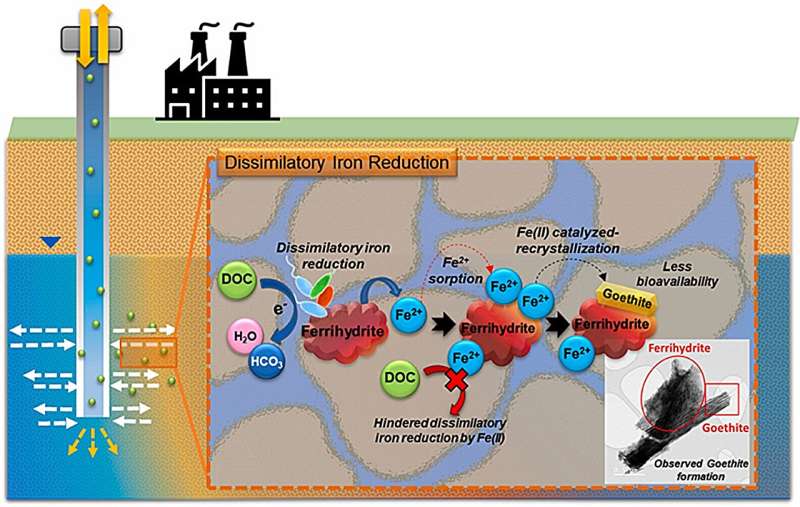
In addition to storing water resources, ASR techniques have the added benefit of improving water quality through various reactions in the ground. The organic pollutants in the recharged water are degraded by the interaction of microorganisms in the aquifer soil with the iron oxide minerals, and in general, the iron oxide minerals are gradually transformed and the effective surface area is reduced, causing the natural attenuation of organic pollutants to stop.
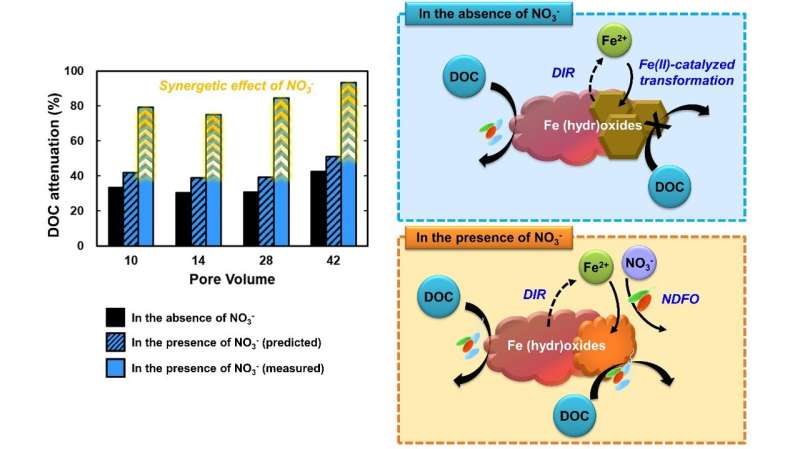
The The Korea Institute of Science and Technology (KIST) researchers found that the coexistence of nitrate in the recharged water leads to the formation of a new type of iron oxide, which results in a much higher removal efficiency than the stoichiometrically predicted organic pollutant removal efficiency.
The coexistence of nitrate increases the duration of natural attenuation because it creates new species of iron oxides that can sustain the degradation of organic contaminants. Furthermore, the researchers found that the pollutant nitrate is removed during the overall reaction.
"This is the first study to confirm the positive role of nitrate in groundwater, which is known only as a water pollutant," said Dr. Seunghak Lee of KIST. "Based on this, we are promoting the development of ASR water quality management protocols that dramatically change the existing water quality management paradigm, such as introducing allowable standards for nitrate residue in the pretreatment process of the recharging water."
More information: Theresia May Anggraini et al, Synergetic effect of nitrate on dissolved organic carbon attenuation through dissimilatory iron reduction during aquifer storage and recovery, Water Research (2023). DOI: 10.1016/j.watres.2023.120954
Journal information: Water Research