This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Applying a small voltage to a catalyst can increase the rates of common reactions used in manufacturing, study finds

A simple technique that uses small amounts of energy could boost the efficiency of some key chemical processing reactions, by up to a factor of 100,000, MIT researchers report. These reactions are at the heart of petrochemical processing, pharmaceutical manufacturing, and many other industrial chemical processes.
The surprising findings are reported in the journal Science, in a paper by MIT graduate student Karl Westendorff, professors Yogesh Surendranath and Yuriy Roman-Leshkov, and two others.
"The results are really striking," says Surendranath, a professor of chemistry and chemical engineering. Rate increases of that magnitude have been seen before but in a different class of catalytic reactions known as redox half-reactions, which involve the gain or loss of an electron. The dramatically increased rates reported in the new study "have never been observed for reactions that don't involve oxidation or reduction," he says.
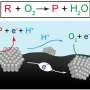
The non-redox chemical reactions studied by the MIT team are catalyzed by acids. "If you're a first-year chemistry student, probably the first type of catalyst you learn about is an acid catalyst," Surendranath says. There are many hundreds of such acid-catalyzed reactions, "and they're super important in everything from processing petrochemical feedstocks to making commodity chemicals to doing transformations in pharmaceutical products. The list goes on and on."
"These reactions are key to making many products we use daily," adds Roman-Leshkov, a professor of chemical engineering and chemistry.
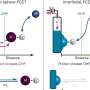
But the people who study redox half-reactions, also known as electrochemical reactions, are part of an entirely different research community than those studying non-redox chemical reactions, known as thermochemical reactions. As a result, even though the technique used in the new study, which involves applying a small external voltage, was well-known in the electrochemical research community, it had not been systematically applied to acid-catalyzed thermochemical reactions.
People working on thermochemical catalysis, Surendranath says, "usually don't consider" the role of the electrochemical potential at the catalyst surface, "and they often don't have good ways of measuring it. And what this study tells us is that relatively small changes, on the order of a few hundred millivolts, can have huge impacts—orders of magnitude changes in the rates of catalyzed reactions at those surfaces."
"This overlooked parameter of surface potential is something we should pay a lot of attention to because it can have a really, really outsized effect," he says. "It changes the paradigm of how we think about catalysis."
Chemists traditionally think about surface catalysis based on the chemical binding energy of molecules to active sites on the surface, which influences the amount of energy needed for the reaction, he says. But the new findings show that the electrostatic environment is "equally important in defining the rate of the reaction."
The team has already filed a provisional patent application on parts of the process and is working on ways to apply the findings to specific chemical processes. Westendorff says their findings suggest that "we should design and develop different types of reactors to take advantage of this sort of strategy. And we're working right now on scaling up these systems."
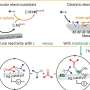
While their experiments so far were done with a two-dimensional planar electrode, most industrial reactions are run in three-dimensional vessels filled with powders. Catalysts are distributed through those powders, providing a lot more surface area for the reactions to take place.
"We're looking at how catalysis is currently done in industry and how we can design systems that take advantage of the already existing infrastructure," Westendorff says.
Surendranath adds that these new findings "raise tantalizing possibilities: Is this a more general phenomenon? Does electrochemical potential play a key role in other reaction classes as well? In our mind, this reshapes how we think about designing catalysts and promoting their reactivity."
Roman-Leshkov adds that "traditionally people who work in thermochemical catalysis would not associate these reactions with electrochemical processes at all. However, introducing this perspective to the community will redefine how we can integrate electrochemical characteristics into thermochemical catalysis. It will have a big impact on the community in general."
While there has typically been little interaction between electrochemical and thermochemical catalysis researchers, Surendranath says, "This study shows the community that there's really a blurring of the line between the two, and that there is a huge opportunity in cross-fertilization between these two communities."
Westerndorff adds that to make it work, "you have to design a system that's pretty unconventional to either community to isolate this effect." And that helps explain why such a dramatic effect had never been seen before. He notes that even their paper's editor asked them why this effect hadn't been reported before.
The answer has to do with "how disparate those two ideologies were before this," he says. "It's not just that people don't really talk to each other. There are deep methodological differences between how the two communities conduct experiments. And this work is really, we think, a great step toward bridging the two."
In practice, the findings could lead to far more efficient production of a wide variety of chemical materials, the team says. "You get orders of magnitude changes in rate with very little energy input," Surendranath says. "That's what's amazing about it."
The findings, he says, "build a more holistic picture of how catalytic reactions at interfaces work, irrespective of whether you're going to bin them into the category of electrochemical reactions or thermochemical reactions." He adds that "it's rare that you find something that could really revise our foundational understanding of surface catalytic reactions in general. We're very excited."
More information: Karl S. Westendorff et al, Electrically driven proton transfer promotes Brønsted acid catalysis by orders of magnitude, Science (2024). DOI: 10.1126/science.adk4902. www.science.org/doi/10.1126/science.adk4902
Journal information: Science
Provided by Massachusetts Institute of Technology
This story is republished courtesy of MIT News (web.mit.edu/newsoffice/), a popular site that covers news about MIT research, innovation and teaching.