This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
proofread
Enrichment of anchoring sites via supramolecular halogen bonds for efficient perovskite nanocrystal LEDs
Colloidal semiconductor nanoparticles can be viewed as a complex of an inorganic single crystal core and a monolayer of organic ligands. The location and type of ligand anchoring on the nanocrystal surface are critical to the nanocrystal morphology, size, bonding patterns, adsorption-desorption processes, and overall stability, optoelectronic properties, etc.
Especially in the perovskite nanocrystals (PNCs) with the nature of soft lattices, the bonding environment of ligand functional groups has played a paramount role in determining the optoelectronic properties and stability of PNCs.
However, the interaction between functional groups and anchoring sites as well as the synergistic and repulsive properties between functional groups are not yet fully understood, which hinders the idealized design of high-performance PNC materials and devices.
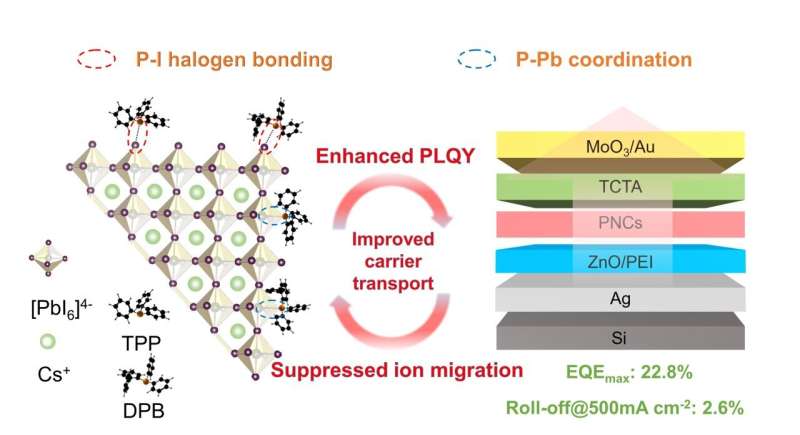
In a recent paper published in Light: Science & Applications, a team of scientists, led by Professor Yu Zhang, from State Key Laboratory of Integrated Optoelectronics and College of Electronic Science and Engineering, Jilin University, China and co-workers have revealed new anchoring sites (supramolecular halogen bonds) on the surface of perovskite nanocrystals (PNCs) by employing the classical triphenylphosphine (TPP) ligand and its derivative 2-(Diphenylphosphino)-biphenyl (DPB).
"It is found that, in addition to the conventionally considered P-Pb coordination interaction, P and I can also form an unexpected halogen bonding interaction." The authors have characterized this in depth by combining nuclear magnetic resonance spectroscopy, Fourier transform infrared spectroscopy (FTIR), and X-ray photoelectron spectroscopy.
"There is a chemical shift in TPP-CsPbI3 compared to TPP, indicating that the P-containing functional groups in TPP interact with the surface of CsPbI3 PNCs, resulting in a change in the coordination environment of P.
"The FTIR spectrum of TPP-passivated PNCs also shows two additional peaks 2 and 3, but they shift to 542 cm-1 and 1120 cm-1, respectively. This suggests that the I...P supramolecular interaction in TPP passivated CsPbI3 PNCs is similar but not identical to that of TPP-I2, which is attributed to the different chemical environment of I atoms in I2 and CsPbI3.
"The Pb 4f spectra of TPP and DPB passivated PNC films shift to the higher binding energy due to the strong binding between the Pb and P functional groups. The I 3d spectra of TPP and DPB passivated PNC films shift to the lower binding energy, which can be considered as the result of the interaction of the nucleophilic atom P in TPP or DPP with the I in PNCs to give electrons to the electrophilic region of I," state the researchers.
The coexistence of the above two types of bonding significantly increased the formation energy of iodine vacancy defects and improved the photoluminescence quantum yield of PNCs. Meanwhile, the direct interaction of P and I enhanced the stability of the Pb-I octahedra and dramatically inhibited the migration of I ions.
In addition, the conjugated nature of benzene rings is also explored, indicating that the introduction of additional benzene rings (DPB) increases the delocalized properties of the PNC surface and significantly improves the charge transport among PNCs.
"Finally, the BPB passivated PNC based top-emitting LEDs achieved a peak EQE of 22.8% and an extremely low efficiency roll-off of 2.6% at the current density of 500 mA cm-2," they added.
"The selection of multifunctional anchoring sites provides a new strategy for improving the optoelectronic properties of PNCs and devices," the scientists forecast.
More information: Po Lu et al, Enrichment of anchoring sites by introducing supramolecular halogen bonds for the efficient perovskite nanocrystal LEDs, Light: Science & Applications (2023). DOI: 10.1038/s41377-023-01266-4
Journal information: Light: Science & Applications
Provided by TranSpread