This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Deciphering crosstalk between bFGF and integrin signaling in maintaining properties of hiPSCs
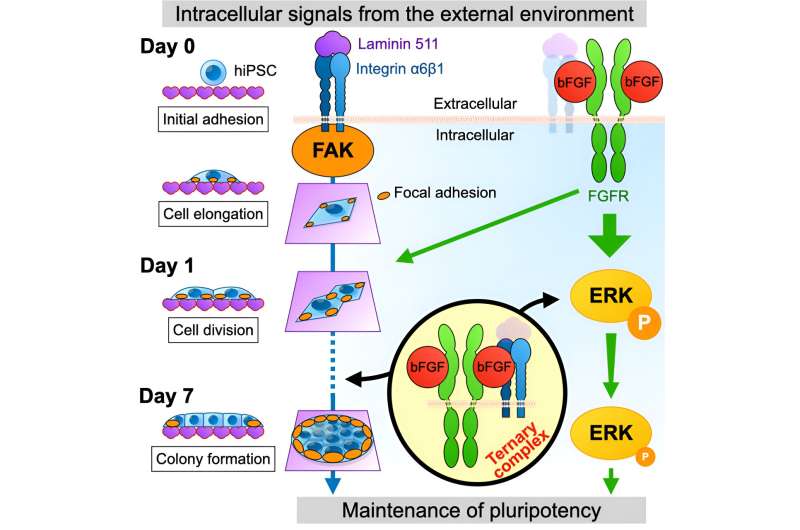
A research team led by Junior Associate Professor Masato Nakagawa (Department of Life Science Frontiers) investigated the effects of basic fibroblast growth factor (bFGF) and its interactions with fibroblast growth factor receptors (FGFRs) and integrins on the maintenance of primed human induced pluripotent stem cell (hiPSC) properties.
The team revealed a new tripartite crosstalk between bFGF, FGFRs, and integrins, shedding light on their roles in maintaining the properties of hiPSCs. The study is published in the journal Regenerative Therapy.
To maintain pluripotency in primed hPSCs, such as induced pluripotent stem cells (hiPSCs) and human embryonic stem cells (hESCs), bFGF and integrins have been known to play separate crucial roles.
Recent studies, however, showed the bFGF-integrin interaction to enhance angiogenesis and tumorigenesis in cancer cell lines, suggesting a potential dual binding function of bFGF to both FGFRs and integrins. In the new study, the researchers identified the importance of bFGF-FGFR and bFGF-integrin binding on the maintenance of hPSC properties.
Specifically, the researchers revealed bFGF to bind to both FGFRs and integrins in hiPSCs, implying the possibility of crosstalk between FGFR and integrin signaling. Using recombinant human bFGF proteins, they demonstrated the direct binding of bFGF to FGFRs and integrins.
The researchers further investigated the physiologic effects of bFGF-FGFR and bFGF-integrin interactions on the maintenance of hiPSC properties by culturing hiPSCs with wild-type bFGF or bFGF mutants that do not bind to FGFRs, integrins, or both.
By examining the expression of pluripotent marker (OCT3/4), they discovered that both bFGF-FGFR and bFGF-integrin interactions were crucial for maintaining the properties of hiPSCs.
To elucidate the crucial period during which bFGF is required, the researchers performed a time course experiment and manipulated the timing of bFGF treatment. They found that bFGF is essential for maintaining the properties of hiPSCs during the initial 24 hours of culture. Notably, they observed bFGF binding to FGFR during this period as critical, whereas binding to integrin was dispensable.
To understand the regulation of the properties of hPSCs, the researchers investigated the downstream events of bFGF-FGFR and bFGF-integrin binding in hiPSCs and focused on extracellular signal-regulated kinases (ERK) and focal adhesion kinase (FAK) activation as downstream signals of bFGF and integrin activation, respectively.
Through this analysis, they revealed that ERK activation triggered by bFGF-FGFR binding was critical for maintaining the properties of hPSCs during the first 24 hours of culture. Conversely, both bFGF-FGFR and bFGF-integrin binding contribute to the accumulation of focal adhesions, composed of activated FAK (pFAK), at the edge of colonies and help maintain hPSC properties.
To investigate whether the simultaneous binding of bFGF to both FGFR and integrin is crucial for maintaining the properties of hPSCs, the researchers examined the effects of treating hiPSCs with a mixture of bFGF mutants—one unable to bind to FGFRs and another unable to bind to integrins—on the maintenance of hPSC properties.
Remarkably, they found that iPSCs treated with this mixture of bFGF mutants could not maintain the properties of hPSCs, suggesting that simultaneous binding of bFGF to both FGFRs and integrins is necessary.
In this study, the researchers found the concurrent binding by bFGF to both FGFR and integrin to be indispensable for maintaining the properties of hPSCs. Moreover, they uncovered the essential role of bFGF-FGFR and bFGF-integrin crosstalk, paving the way for further work on a greater understanding of the molecular mechanisms involved in pluripotency maintenance in hiPSCs.
More information: Yu-Shen Cheng et al, Simultaneous binding of bFGF to both FGFR and integrin maintains properties of primed human induced pluripotent stem cells, Regenerative Therapy (2023). DOI: 10.1016/j.reth.2023.12.008
Provided by Kyoto University