This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers decode aqueous amino acid's potential for direct air capture of CO₂
Scientists at the Department of Energy's Oak Ridge National Laboratory have made a significant stride toward understanding a viable process for direct air capture, or DAC, of carbon dioxide from the atmosphere. This DAC process is in early development with the aim of achieving negative emissions, where the amount of carbon dioxide removed from the envelope of gases surrounding Earth exceeds the amount emitted.
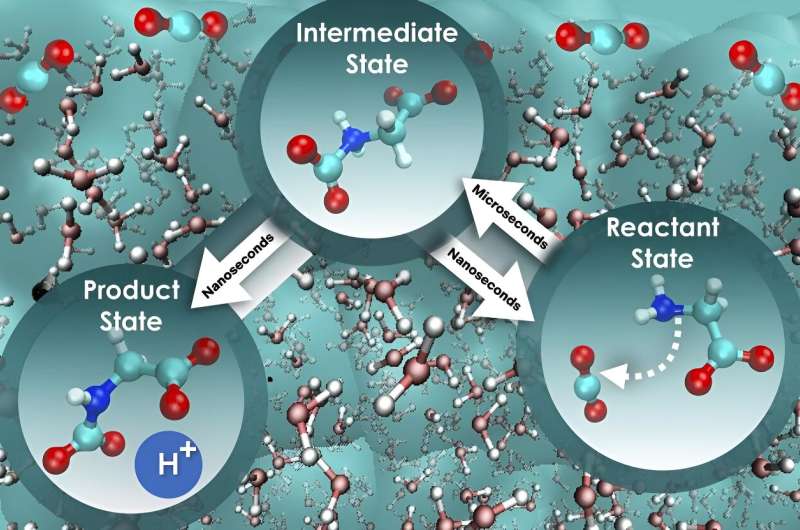
The research, recently published in Cell Reports Physical Science, focused on the foundational steps of carbon dioxide sequestration using aqueous glycine, an amino acid known for its absorbent qualities. By combining a series of advanced computational methods, the scientists probed less-explored dynamic phenomena in liquid solutions related to the rate at which carbon dioxide can be captured.
"Chemical reactions in water are complicated, especially when the motion of water molecules plays a big role," said Santanu Roy, who designed the computational investigation with colleague Vyacheslav Bryantsev. "Water molecules and chemicals engage in something similar to a coupled dance that can marginally or significantly slow the reaction. Understanding these dynamic interactions, known as nonequilibrium solvent effects, is essential to getting the full picture of how reactions work and how fast they happen."
The researchers discovered that when examining the rate at which carbon dioxide is absorbed, focusing solely on the free energy barrier—the energy threshold that must be overcome for a system to transition from one state to another—is an oversimplification that does not provide the full picture. This incomplete approach can lead to an inaccurate understanding of reaction kinetics, the factors that influence the speed at which a reaction occurs.
"We employed a more complete approach that considers the influence of water on the motion along the reaction path, and the outcome was intriguing," Bryantsev said. "The initial step, where glycine interacts with carbon dioxide, is nearly 800 times slower compared with the next step, where a proton is released to ultimately form a mixture of product state for holding the absorbed carbon dioxide.
"Strikingly, the free energy barrier remains constant for both steps, and so this different perspective truly sets the speed of these two critical stages apart and offers a pathway to boost the efficiency of carbon dioxide absorption and separation."
The extensive ab initio molecular dynamics simulations used in this study were still limited by their short time and length scales and high computational costs in representing the chemical reactions.
"For future projects, we intend to combine the emerging machine-learning approach with highly accurate simulations and develop interatomic interaction potentials based on deep neural networks. This will allow us to perform molecular simulations with high accuracy at large scales with significantly reduced computational costs," said Xinyou Ma, who carried out the simulations.
Roy added, "While we have portrayed a molecular-level kinetics picture of carbon dioxide capture by aqueous amino acids, accessing large length and time scales through the use of the machine-learning approach will help us understand the effects of macroscopic factors such as temperature, pressure and viscosity on DAC and how these effects are related to the attained molecular picture."
Overall, the study's findings shed light on the intricate workings of DAC and emphasize the vital role of kinetics, thermodynamics and molecular interactions in removing carbon dioxide from the atmosphere by aqueous amino acids. As these mechanisms become more accurately understood, the prospect of deploying a large-scale DAC technology will become more feasible. Worldwide, several different DAC projects are in various stages of research, testing and development.
More information: Xinyou Ma et al, An ab initio free energy study of the reaction mechanism and rate-limiting steps of CO2 capture by aqueous glycine, Cell Reports Physical Science (2023). DOI: 10.1016/j.xcrp.2023.101642
Journal information: Cell Reports Physical Science
Provided by Oak Ridge National Laboratory