This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers develop new method to analyze proteins in ecologically significant moss

Though the changing climate threatens the world's vegetation, a type of peat moss has surprised scientists with its resilience: Sphagnum divinum is actively evolving in response to hot, dry conditions.
Researchers at the Department of Energy's Oak Ridge National Laboratory developed resources, including a database with all of S. divinum's proteins and a method to determine their functions, in order to illuminate how S. divinum adapts under rapidly changing climate conditions. Their work has been published in Bioinformatics.
Sphagnum mosses dominate peatlands that store about a third of the world's terrestrial carbon. As environmental stressors diminish these reserves, the peat releases carbon into the atmosphere as a greenhouse gas, further accelerating the pace of warming. Understanding the genetic mechanisms that enable resilience under changing conditions is vital to inform strategies to maintain or enhance carbon storage in peat bogs.
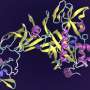
"Think of an organism as an engine, which works because of the way that its components fit together in three dimensions," said Ada Sedova, a computational biologist at ORNL. "Proteins are like the individual parts. Their structure determines their function, so knowing their shape helps us predict what they do for the machine as a whole."
To reveal the roles of proteins, genetic code can be modeled structurally. ORNL researchers previously streamlined this process with high-performance computing resources. They modified DeepMind's AlphaFold—an AI-powered tool that predicts a protein's 3D shape from its genetic sequence—to run on Summit, a supercomputer housed at ORNL's Oak Ridge Leadership Computing Facility. The project predicted structures for S. divinum's 25,134 proteins at unprecedented speeds.
The team also developed a technique to compare proteins by aligning them with one another. Using this procedure, they found differences between two similar Sphagnum proteins. One is likely an enzyme that aids a biological process, whereas the other could serve a regulatory function such as managing the organism's heat stress response.
"This high-throughput method can scale to large computing clusters," said Sedova, "which addresses the data challenges that developed when proteome-scale structure prediction with AlphaFold became a reality."
This work supplements ORNL's ongoing research about Sphagnum moss, including the SPRUCE whole-ecosystem manipulation experiment and a project that previously sequenced the genomes of two species.
"Determining the function of proteins in a laboratory setting is a time-consuming and frequently unsuccessful endeavor. Advancing computational methods for delivering reliable functional predictions will significantly expedite the discovery of molecular genetic processes that contribute to climate resilience," said Dave Weston, a molecular plant physiologist at ORNL.
Though most proteins have a set form and function, some lack this rigidity. Disordered proteins give an organism flexibility, allowing it to respond to a dynamic environment. "In our research, AlphaFold indicated that around 30% of S. divinum's proteins lack a defined structure," said Sedova. "This suggests that disordered proteins could be critical for heat, stress and drought response."
Going forward, the database and analytical method developed at ORNL will support biologists looking to understand the essential compounds that make up Sphagnum mosses. This information will help reveal how plants respond to a changing world.
More information: Russell B Davidson et al, Predicted structural proteome of Sphagnum divinum and proteome-scale annotation, Bioinformatics (2023). DOI: 10.1093/bioinformatics/btad511
Journal information: Bioinformatics
Provided by Oak Ridge National Laboratory