This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Protein coding gene coordinates cell division, protein synthesis, and mitochondrial activity in β-cells
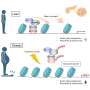
Pancreatic β-cells are essential for secreting insulin to regulate glucose homeostasis. During metabolic stress they respond by increasing insulin production and secretion and proliferation to meet the increased metabolic demand. A detailed understanding of this process is important to understand the processes leading to β-cell decompensation and demise in obesity and type 2 diabetes.
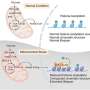
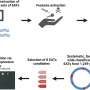
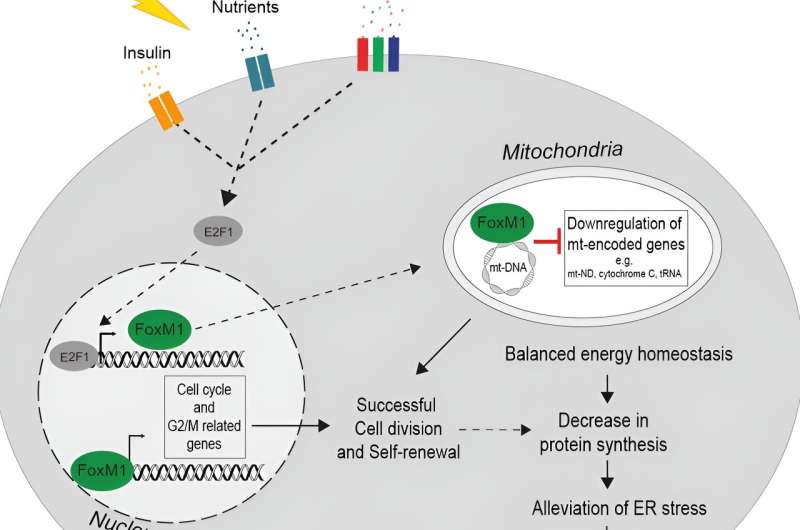
Researchers at IMHS characterize β-cell responses to acute metabolic stress in vivo by employing RNA-seq, ATAC-seq, scRNA-seq, ChIP-seq and real-time imaging to decipher temporal events of chromatin remodeling and gene expression regulating the unfolded protein response (UPR), protein synthesis, mitochondrial function, and cell cycle progression.
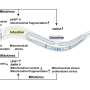
They identified proliferation-prone pancreatic β-cells with enhanced UPR and decreased protein synthesis and insulin secretion during metabolic stress. Insulin depletion and alleviation of ER stress precede the progression of the cell cycle and ensure appropriate insulin synthesis. FoxM1 mediates this process by regulating translation and mitochondrial activity by repressing mitochondrial genes. The study is published in the journal Cell Reports.
More information: Ahmad Kobiita et al, FoxM1 coordinates cell division, protein synthesis, and mitochondrial activity in a subset of β cells during acute metabolic stress, Cell Reports (2023). DOI: 10.1016/j.celrep.2023.112986
Journal information: Cell Reports
Provided by ETH Zurich