This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers improve fitness of cells used in cell transplants
A readily available, inexpensive, small molecule drug can improve the fitness of hematopoietic stem and progenitor cells (HSPCs) that are modified outside the body, potentially improving the success of procedures such as ex vivo gene therapy, according to a new study by researchers at Children's Hospital of Philadelphia (CHOP).
The study, published in the journal Blood, showed that targeting components in the cells called extracellular vesicles (EVs) relieves the stress on cells when outside of the body, improving their performance when they are transplanted back inside.
"We initially embarked on this project to better understand the role of these vesicles, assuming—wrongly, as it turns out—that blocking their function would have detrimental effects on the cells," said first author Stephanie Hurwitz, MD, Ph.D., a research fellow in the Kurre Lab at CHOP and Hematopathologist at the University of Pennsylvania.
"We discovered the opposite is true: blocking the formation of these vesicles over a relatively short period of time actually improved the cells' fitness, initiating an adaptive stress response that protects them from the stress of being outside of the body. Ultimately, this serendipitous finding could enhance the quality of transplant products used in treatments like ex vivo gene therapy, improving patient outcomes."
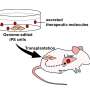
Hematopoietic stem cell transplantation (HSCT) involves the transfer of HSPCs from a donor to patients with both benign and malignant diseases. In some cases, as in ex vivo gene therapy, the patient acts as their own donor: the patient's HSPCs are removed, modified outside of the body using gene addition or gene editing, and then transplanted back in. When enough cells are available for modification, this strategy has proven to be a powerful platform for treating genetic blood disorders, including sickle cell disease and beta thalassemia.
However, maintaining the fitness of these cells outside of the body poses a challenge, and any loss of fitness introduces the risk that the cells will not properly restore the blood and immune cell system in patients. For this reason, researchers have sought to better understand HSPC programming and signaling so that every attempt could be made to maintain cell health ahead of transplant.
One process that we now learn contributes to cell equilibrium is the secretion of EVs, which are important for delivering messages from cell to cell and maintaining proper cell function. However, technical challenges have previously prevented researchers from fully understanding the role these EVs might play in HSPC fitness outside of the body.
To explore the role of EVs, the researchers exposed both mouse and human HSPCs to GW4869, a readily available small molecule drug that blocks neutral sphingomyelinase-2 (nSMase2), an enzyme involved in EV formation. The researchers found that an exposure of 24 hours led to sustained improvements in cell fitness and transplantation efficiency after ex vivo culture, with improved rates of blood cell function in mouse models.
Additionally, the researchers found that blocking nSMase2 in human CD34+ cells leveled disparities in how well the cells of varying HLA types performed after transplantation into mice. This is of particular interest given important role of HLA in long-term transplantation outcomes.
Drilling down on the mechanisms behind these phenomena, the researchers determined that blocking nSMase2 activates an integrated stress response, effectively putting the cells into a state of quiescence, where they are protected from external stressors. This occurs due, in part, from GW4869 disrupting sphingolipid metabolism, which impairs the release of nSMase2-dependent EVs.
"While numerous advances have been made over past decades to improve safety and efficiency of HSPC transplantation and ex vivo gene therapy, constraints in cell quantity and graft success continue to limit outcomes," said senior author Peter Kurre, MD, Director of the Pediatric Comprehensive Bone Marrow Failure Center at CHOP.
"This important study, which links EV trafficking and the integrated stress response as a regulatory dyad guarding HSPC integrity and long-term fitness, has important implications for potentially broadening patient eligibility to ex vivo gene therapies. Researchers should continue to explore the potential of this small molecule drug in improving ex vivo HSPC culture and transplantation."
More information: Stephanie N Hurwitz et al, Neutral sphingomyelinase blockade enhances hematopoietic stem cell fitness through an integrated stress response, Blood (2023). DOI: 10.1182/blood.2023022147
Journal information: Blood
Provided by Children's Hospital of Philadelphia