This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Biological origami at molecular level: Cytosolic interactome protects against protein unfolding
Human cells are protecting their proteins from unfolding and aggregating. That's what biophysicist Alireza Mashaghi and his team discovered after seven years of in-depth research into the folding mechanisms of proteins. With an unprecedented approach, the team was able to study the folding of a single protein molecule.
Proteins are fundamental building blocks of life. These complex molecules do most of the work in our cells. To do their job properly, each protein must be folded into a specific structure. And that's where things sometimes go wrong. If proteins somehow unfold, for example, because of disease, they lose their function or lump together. Understanding the mechanisms behind the folding and stability of proteins is therefore crucial for developing potential new treatments.
How does a protein unfolds under stress?
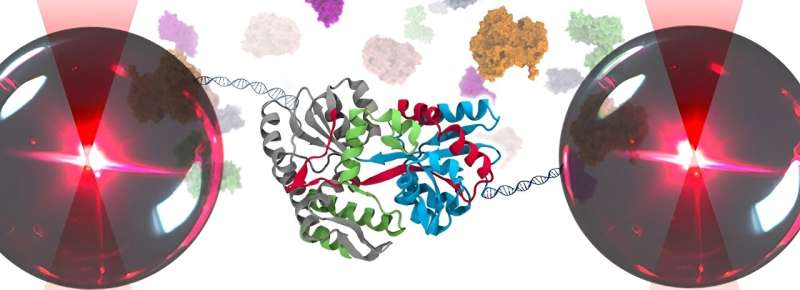
"We wanted to observe how a single protein unfolds when it's experiencing stress in the cellular environment," explains Barbara Scalvini, one of the researchers. She and her colleagues developed a new approach combining optical tweezers and circuit topology: a system to determine and classify the shape of proteins. The findings have been published in Advanced Biology.
With this, they studied a protein in a cytosolic solution—a close approximation of the complex cytoplasmic matrix found within living cells. "This allowed us firstly to explore how the cellular environment influences the unfolding pathway of a protein. And secondly, to evaluate the resulting protein shape," Scalvini said.
Protective powers
The researchers unveiled a previously unknown protective effect of the cellular environment: it seems to protect the proteins against unfolding and aggregating. "We used the optical tweezers to pull the proteins by their ends, mimicking natural unfolding. We noticed that a part of it would stay folded, and that we needed more force to completely unfold it. This suggests that the molecular interactions within the cell have a stabilizing effect," said Scalvini.
Group leader, Alireza Mashaghi, said, "When a cell experiences stress, a protein can unfold to a completely unfolded chain. Once that has happened, it's very hard to reverse. But we noticed the cytoplasm puts a break on this process, not allowing the unfolding to go all the way. This protects the proteins and ensures a proper functionality, and also makes it easier for proteins to refold once the stress in resolved."
Mashaghi calls the study a significant advancement in the field of biophysics. "We are unraveling the intricate relationship between protein folding and the cellular environment. Our results pave the way for future studies in more realistic biological environments. This will help us bridge the gap between lab studies and the real world; between fundamental biophysics and medicine."
'Making the cell environment work in our favor'
The findings hold immense promise for numerous areas of research. Mashaghi says, "Think of drug development and understanding the mechanisms of various diseases caused by protein misfolding, such as neurodegenerative disorders or muscular dystrophies. With further research, we may be able to harness the protective properties of the cell's environment."
More information: Barbara Scalvini et al, Cytosolic Interactome Protects Against Protein Unfolding in a Single Molecule Experiment, Advanced Biology (2023). DOI: 10.1002/adbi.202300105
Alireza Mashaghi et al, Alternative modes of client binding enable functional plasticity of Hsp70, Nature (2016). DOI: 10.1038/nature20137
Journal information: Nature
Provided by Leiden University