How do cellular machines unfold misfolded proteins?
Protein chains typically fold to function. Folding is a complex process and if done correctly leads to a unique functional fold topology for a given protein chain. Other topologies are also possible but are often non-functional or toxic. These misfolded proteins are then unfolded and subsequently refolded to the correct fold topology; otherwise, they undergo degradation.
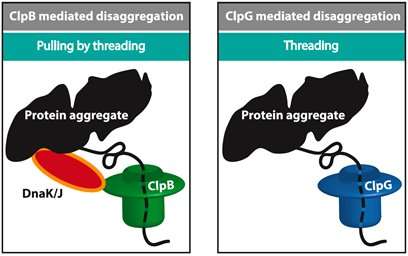
Several machines including ClpB and ClpG are responsible for unfolding a folded protein. ClpB works closely with HSP70 (DnaK) and HSP40 (DnaJ) and uses energy to unfold a chain while ClpG does not depend on HSP70. A major question is that why cells are equipped with different types of machines and what determines the efficiency of unfolding. Alireza Mashaghi and his team at LACDR/Leiden University solved this puzzle by monitoring unfolding of misfolded chain models at the single-molecule level. Three unfolding approaches were compared, namely, threading through a pore, pulling from the ends, and pulling by threading.
The results of this analysis, which are published on October 25th in the Journal of Physical Chemistry B, reveal that circuit topology of the folded chain critically determines the number of pathways and the efficiency of unfolding in a manner that depends on the employed mechanical approach. The study provides insights into cellular protein unfolding mechanisms. These findings may help in selecting optimal chaperone targets for pharmacotherapy of misfolding diseases.
More information: Narges Nikoofard et al. Implications of Molecular Topology for Nanoscale Mechanical Unfolding, The Journal of Physical Chemistry B (2018). DOI: 10.1021/acs.jpcb.8b09454
Journal information: Journal of Physical Chemistry B
Provided by Leiden University