This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers investigate how the cellular environment affects protein conformational dynamics
Protein dynamics through motions of loops, linkers, and hinges can generate distinctive conformations that are important for protein function. Most proteins perform their functions in cells. However, how the complex cellular environment affects the conformational dynamics of proteins remains unclear.
Recently, a research team from the Qingdao Institute of Bioenergy and Bioprocess Technology (QIBEBT) of the Chinese Academy of Sciences (CAS) has investigated the protein loop conformational dynamics in Escherichia coli (E.coli) cells by Nuclear Magnetic Resonance (NMR) Spectroscopy. Their findings were published in Science Advances on July 21.
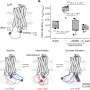
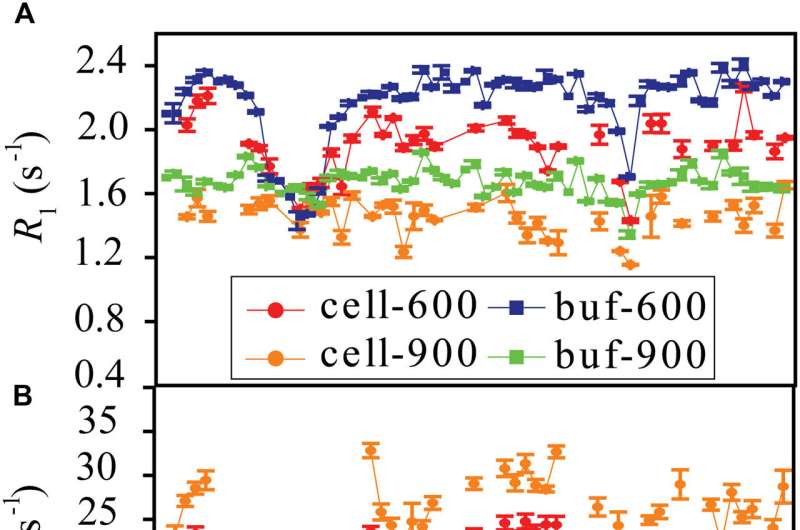
The researchers found that weak interactions between the protein and surrounding macromolecules in E.coli cells hindered protein rotational diffusion, which extended the dynamic detection timescale up to microseconds by the NMR spin relaxation method. The loop picosecond to microsecond dynamics was confirmed by nanoparticle-assisted spin relaxation and residual dipolar coupling methods.
Further investigation characterized the protein loop 1 region with strong flexibility through the sequence parameter S2 (0 represents the most flexible, and 1 represents the most rigid), and the linkers between α1 and β3 also showed significant flexibility. The conformational dynamics of intracellular proteins were consistent with the results obtained in aqueous solutions, but with significant differences in numerical values.
The loop interactions with the intracellular environment were perturbed through point mutation of the loop sequence. For the sequence of the protein that interacted stronger with surrounding macromolecules, the loop became more rigid in cells. In contrast, the mutational effect on the loop dynamics in vitro was small.
This study realized direct measurement of conformation dynamics of protein loop in cells, and provided direct evidence of how the cell environment changes the conformation dynamics of protein loop through weak interaction, which may affect the function of proteins. It also highlights the importance of directly studying protein properties and functions within living cells.
More information: Mengting Wang et al, Intracellular environment can change protein conformational dynamics in cells through weak interactions, Science Advances (2023). DOI: 10.1126/sciadv.adg9141
Journal information: Science Advances
Provided by Chinese Academy of Sciences