This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
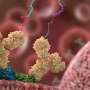
Scientists use sound waves to test how well synthetic antibodies bind to their antigen targets

We rely on temporary protein-protein bonds for essential processes including enzymatic reactions, antibody binding, and response to medication. Being able to accurately characterize these bonds is important for testing the performance of potential therapies, but the currently available methods to do so are limited in their abilities either to provide information at the single bond level or to test large numbers of bonds.
On June 7th in Biophysical Journal, researchers presented a more accessible method to measure the strength and duration of protein-protein bonds under similar loads to those they would experience inside our bodies. The method uses sound waves to pull bonded proteins apart and DNA leashes to keep the two proteins close together so that they can re-bond after their connection is ruptured. This innovation allows the same protein bonds to be re-tested up to 100 times, providing valuable information about how bond strength changes as molecules age.
"We wanted to propose a method that is sufficiently modular to apply to different type of bonds, that has a reasonable throughput, and that reaches high molecular precision that is currently only available with very refined techniques, like optical or magnetic tweezers, that are often difficult to grasp for non-specialists," says senior author Laurent Limozin, a biophysicist at the Center National de la Recherche Scientifique (CNRS).
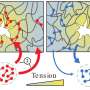
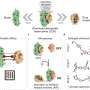
To do this, Limozin's team, in collaboration with colleagues from Marseille and Paris, combined two existing technologies: acoustic force spectroscopy, which allows many molecular pairs to be tested simultaneously, and DNA scaffolds, which enable the same bonds to be tested repeatedly.
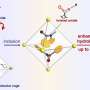
During acoustic force spectroscopy, pairs of bonded proteins are tested inside a liquid-filled chamber. The proteins are restrained by DNA scaffolding such that one strand of DNA attaches the first protein to the bottom of the chamber, while another strand attaches the second protein to a small silica bead. When the researchers blast the chamber with a soundwave, the wave's force pulls the silicon bead—and the protein it's attached to—away from the bottom of the chamber. If the force is strong enough, this pulling action ruptures the bond between the two proteins. However, in this new method, a third strand of DNA acts as a leash to keep the proteins close together after their bond is ruptured.
"The originality of our method is that in addition to these two strands on each side, in the middle you have this leash that connects the two strands and keeps the proteins together upon rupture," says Limozin. "Without this leash, the detachment would be irreversible, but this allows you to repeat the measurement almost as many times as you wish."
As a proof of concept, the research team used the technique to characterize two single-molecule interactions of biomedical interest—the bond between proteins and rapamycin, an immunosuppressive drug, and the bond between a single-domain antibody and an HIV-1 antigen.
The researchers observed these cycles of bonding and rupturing using a microscope. Being able to test the same protein-protein bond multiple times is important for exploring variation between molecularly identical pairs. It also allows researchers to examine how these interactions change as the molecules age, which could be important for determining the half-life of drugs or antibodies.
"With this tool we have a way to go deeper and really probe experimentally ideas about molecular heterogeneity and molecular aging," says Limozin. "We, and others, suspect that characterizing these properties will be very useful for designing future therapeutics that will need to work in situations where mechanical forces are involved."
More information: Combining DNA scaffolds and acoustic force spectroscopy to characterize individual protein bonds, Biophysical Journal (2023). DOI: 10.1016/j.bpj.2023.05.004
Journal information: Biophysical Journal
Provided by Cell Press