This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Resolving hydrogen atoms in small organic molecules depending on chemical bond type
Extremely intense X-ray pulses can determine the positions of some hydrogen atoms in organic molecules that form small crystals, an all-RIKEN team has shown. Many areas, including drug discovery and materials research, stand to benefit from this demonstration.
Ever since William Lawrence Bragg and his father William Henry Bragg demonstrated that X-rays scattered from crystals produce distinctive patterns 110 years ago, X-ray diffraction has been the technique of choice for determining the structure of crystalline materials. However, many materials form crystals that are too small to be analyzed by X-ray diffraction.
"Because many compounds cannot be obtained in large crystals, the ability to analyze the structures of small crystals is important in fields such as synthetic organic chemistry, pharmaceutical science and materials science," says Koji Yonekura of the RIKEN SPring-8 Center.
Switching from X-rays to electrons can allow the structures of smaller crystals to be determined, but it has the downside that it requires very thin samples.
Another promising approach is to use the ultra-intense, ultrashort X-ray pulses produced by X-ray free electron lasers (XFELs)—large facilities that are kilometers long.
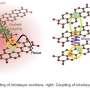
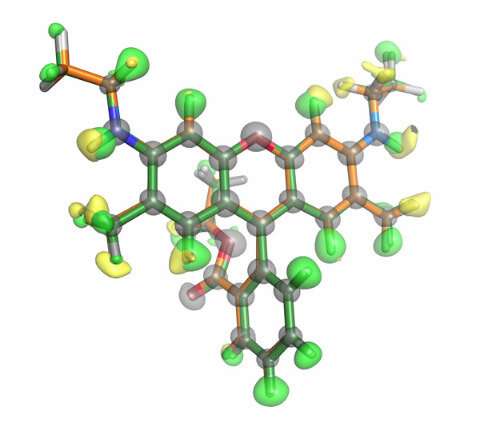
Now, Kiyofumi Takaba, Saori Maki-Yonekura, Yonekura and co-workers have used X-rays from an XFEL to determine the structure of a small organic molecule, a fluorescent dye called rhodamine-6G. They also compared the results with those obtained using 3D electron diffraction for the same molecule.
Using both techniques, the researchers were able to resolve the positions of some of the hydrogen atoms, which is remarkable since they are the smallest atom, consisting of just a proton and electron. The positions of the hydrogen atoms resolved by the two techniques depended on the type of bonds between hydrogen atoms. Their work is published in the journal Nature Chemistry.
"This is the first time anyone has demonstrated that hydrogen atoms in small crystals of organic compounds can be visualized by XFEL diffraction," notes Takaba. "It is an important achievement because the position of a hydrogen atom reveals the polarity of chemical bonds and because hydrogen atoms can significantly affect an organic molecule's properties and functions."
While the molecular structures obtained by the two techniques were very similar to each other, they also provided complimentary information since electrons and X-rays interacted differently with the samples. "XFEL and electron crystallography can reveal different and detailed features of organic compounds," says Maki-Yonekura. Specifically, XFEL could determine atomic coordinates more accurately, whereas electron diffraction was more sensitive to the distribution of electric charges in the molecule.
Importantly, no special sample preparation was needed for either method, making them practical to perform.
The team intends to continue to explore how specimens interact with different probes. "We expect that this will extend not only the application of these techniques but also our theoretical understanding of the techniques themselves," says Takaba.
More information: Kiyofumi Takaba et al, Structural resolution of a small organic molecule by serial X-ray free-electron laser and electron crystallography, Nature Chemistry (2023). DOI: 10.1038/s41557-023-01162-9
Journal information: Nature Chemistry
Provided by RIKEN