This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Protein nanoparticle vaccine with adjuvant improves immune response against influenza, find researchers
A novel type of protein nanoparticle vaccine formulation containing influenza proteins and adjuvant to boost immune responses has provided complete protection against influenza viral challenges, according to a new study published by researchers in the Institute for Biomedical Sciences at Georgia State University.
The findings published in the journal Small describe a promising influenza vaccine candidate that uses adjuvants, substances that increase immune response to a vaccine, to boost effectiveness against viral infections.
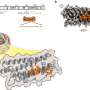
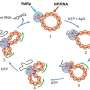
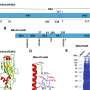
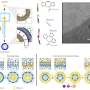
The researchers developed a novel type of core/shell protein nanoparticle consisting of influenza nucleoprotein as the core and NA1-M2e or NA2-M2e surface proteins as the coating antigens. In the new nanoparticle fabrication, the surface protein coating can be precisely controlled and any excessive coating proteins can be recovered for reuse.
Thus, nanoparticle quality and yield are improved significantly. Mice were intramuscularly or intranasally vaccinated with the resulting protein nanoparticles with and without immune-stimulating complexes as adjuvants to determine the immune response and protective efficiency against influenza viral infections.
"We found that the novel protein nanoparticles combined with the adjuvants could induce significantly improved mucosal immune responses and the accumulation of lung resident memory cells in the local respiratory tracts, providing complete protection against influenza viral infections," said Dr. Wandi Zhu, first author of the study and a research assistant professor in the Institute for Biomedical Sciences at Georgia State.
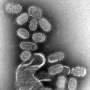
Influenza A virus is one of the most threatening respiratory pathogens and can cause severe morbidity, mortality and heavy economic burdens, especially in flu epidemics or pandemics. While vaccination is effective in preventing or reducing viral infections during annual flu seasons, the selection of vaccine strains depends on circulating viral surveillance and prediction.
Mismatched strains could significantly impair vaccine efficiency. Also, the production of the current quadrivalent influenza vaccine is time-consuming. New vaccine technologies are needed to easily manufacture a universal influenza vaccine. The researchers have focused their work on developing different types of protein nanoparticle vaccines against both influenza A and influenza B viral infections.
"The epidemics caused by the influenza virus seriously threaten public health and the economy," said Dr. Baozhong Wang, senior author of the study and Distinguished University Professor in the Institute for Biomedical Sciences at Georgia State. "Adding appropriate adjuvants to improve immunogenicity and finding effective mucosal vaccines to combat respiratory infection at the portal of virus entry are important strategies to boost protection."
Adjuvanted protein nanoparticles could improve strong systemic and mucosal immune responses conferring protection in different immunization routes. This work highlights the importance of applying adjuvants in mucosal vaccine formulations. The adjuvanted protein nanoparticles can be used as mucosal vaccines alone or in combination with other vaccines to improve mucosal immunity and protection in the future, Zhu explained.
"The new surface protein coating is a more controlled and efficient fabrication method than our previous process, significantly increasing the utilization of the initial proteins," Zhu said. "The technique could be used to develop universal influenza vaccines consisting of influenza antigens from influenza A and influenza B viruses. The combination of the vaccine with appropriate adjuvants will be promising mucosal vaccine candidates."
More information: ISCOMs/MPLA-Adjuvanted SDAD Protein Nanoparticles Induce Improved Mucosal Immune Responses and Cross-Protection in Mice, Small (2023). DOI: 10.1002/smll.202301801. onlinelibrary.wiley.com/doi/10.1002/smll.202301801
Journal information: Small
Provided by Georgia State University