This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
In a first, researchers capture fleeting 'transition state' in ring-shaped molecules excited by light
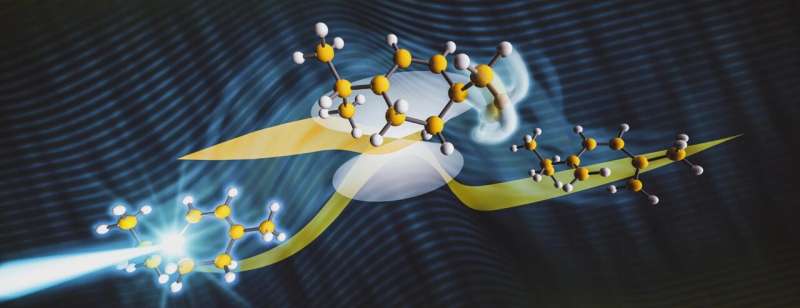
Using a high-speed "electron camera" at the Department of Energy's SLAC National Accelerator Laboratory and cutting-edge quantum simulations, scientists have directly imaged a photochemical "transition state," a specific configuration of a molecule's atoms determining the chemical outcome, during a ring-opening reaction in the molecule α-terpinene. This is the first time that scientists have precisely tracked molecular structure through a photochemical ring-opening reaction, triggered when light energy is absorbed by a substance's molecules.
The results, published in Nature Communications, could further our understanding of similar reactions with vital roles in chemistry, such as the production of vitamin D in our bodies.
Transition states generally occur in chemical reactions which are triggered not by light but by heat. They are like a point of no return for molecules involved in a chemical reaction: As the molecules gain the energy needed to fuel the reaction, they rearrange themselves into a fleeting configuration before they complete their transformation into new molecules.
"Transition states really tell you a lot about how and why reactions happen," said co-author and SLAC scientist Thomas Wolf. "The investigation of similar critical configurations in photochemical reactions could lead to a better understanding of reactions with key roles in chemistry and biology. It's important that we can now look at some specific characteristics of such reactions using our diffraction techniques."
Until now, no method existed that was sensitive enough to capture these fleeting states, which last for only millionths of a billionth of second. At MeV-UED, SLAC's instrument for ultrafast electron diffraction, the researchers sent an electron beam with high energy, measured in millions of electronvolts (MeV), through a gas to precisely measure distances between the atoms within the molecules in the gas. Taking snapshots of these distances at different intervals after an initial laser flash allows scientists to create a stop-motion movie of the light-induced atomic rearrangements in the molecules.
"These reactions are important for understanding the quantum mechanics underpinning photochemistry," said SLAC scientist and co-author Yusong Liu. "Comparing our experimental results with quantum simulations of the reaction allows us to get a highly accurate picture of how molecules behave and benchmark the predictive power of theoretical and computational methods."
In a previous study of a related reaction, MeV-UED allowed the team to capture the coordinated dance between electrons and nuclei. The results provided the first direct confirmation of a half-century-old set of rules about the final product's stereochemistry, or the three-dimensional arrangement of its atoms.
In the present experiment, the researchers discovered that some parts of the atomic rearrangements happen earlier than other parts, which provides an explanation for why the specific stereochemistry is created by the reaction.
"I recently looked back on some old presentations I did in college about these types of reactions and the famous set of rules that predict the outcomes. But these rules don't actually explain why and how reactions happen." Wolf said. "And now I'm coming back to that and can start answering these questions and that makes it incredibly exciting for me."
Another big motivation for doing these experiments, Wolf said, is that the same reaction also happens in biological processes such as the biosynthesis of vitamin D in human skin. The researchers plan to conduct follow-up studies further exploring this connection.
More information: Y. Liu et al, Rehybridization dynamics into the pericyclic minimum of an electrocyclic reaction imaged in real-time, Nature Communications (2023). DOI: 10.1038/s41467-023-38513-6
Journal information: Nature Communications
Provided by SLAC National Accelerator Laboratory




















