This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Going small and thin for better hydrogen storage
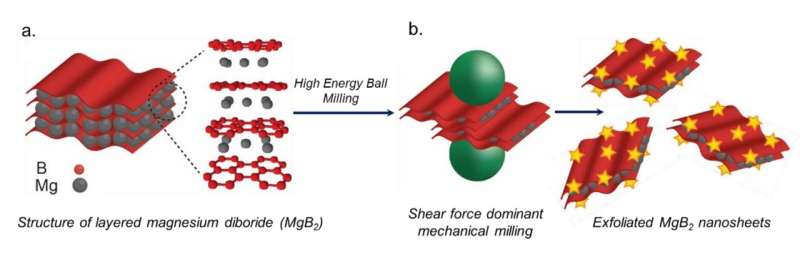
A collaboration including scientists from Lawrence Livermore National Laboratory (LLNL), Sandia National Laboratories, the Indian Institute of Technology Gandhinagar and Lawrence Berkeley National Laboratory has created 3-4 nanometer ultrathin nanosheets of a metal hydride that increase hydrogen storage capacity. The research appears in the journal Small.
There is a need for sustainable energy storage technologies that can address the intermittent nature of renewable energy resources. Hydrogen-based technologies are promising long-term solutions that reduce greenhouse gas emissions.
Hydrogen has the highest energy density of any fuel and is considered a viable solution for ground transportation, aircraft and marine vessels. However, hydrocarbon fuel sources outperform compressed hydrogen gas in terms of volumetric energy density, motivating the development of alternative, higher-density materials-based storage methods.
Complex metal hydrides are a class of hydrogen storage materials that while having high absolute storage capacity, can require extreme pressures and temperatures to achieve that capacity. The team tackled this challenge by nano-sizing, which increases the surface area to react with hydrogen and decreases the required depth of hydrogenation. Previous studies have analyzed nanoscale magnesium diboride (MgB2), including work by LLNL, however, the material in that study was not as thin and wound up clustering together.
The material created in this most recent collaboration came from solvent-free mechanical exfoliation in zirconia, yielding material that is only 11-12 atomic layers thick and can hydrogenate to about 50 times the capacity of the bulk material.
This 50-fold increase in the hydrogenation neatly corresponds to a 50-fold increase in the surface to volume ratio, suggesting that both the bulk and nanosheet material hydrogenate approximately the first two layers, a universal behavior independent of particle size. For two layers on either side of the 11-12-layer nanomaterial, this represents a third of the maximum hydrogen capacity of MgB2.
MgB2 consists of alternating magnesium and boron layers for which charge transfer from the magnesium layer to the boron layer drives the boron layer stability. LLNL calculations reveal that the incomplete Mg coverage on the surface of the material energetically favors a surface structure with islands of complete magnesium coverage and other areas of less stable disordered surface boron layers. Building from previous work on the disordering of surface boron layers, calculations show how magnesium coverage on MgB2 evolves as it hydrogenates.
"These results show how a reactive MgB2 surface with exposed boron may become more stable as it hydrogenates because the magnesium coverage increases," said LLNL physicist and author Keith Ray. "By this mechanism the hydrogenation slows and halts for moderate hydrogenation conditions.
"Further nano-sizing or a novel chemical modification to delay or disrupt the increase in surface magnesium may further increase MgB2 performance as a hydrogen storage material," he added.
More information: Harini Gunda et al, Hydrogen Storage in Partially Exfoliated Magnesium Diboride Multilayers, Small (2022). DOI: 10.1002/smll.202205487
Journal information: Small
Provided by Lawrence Livermore National Laboratory