This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Engineer discovers method to improve pharmaceuticals through dolphin research
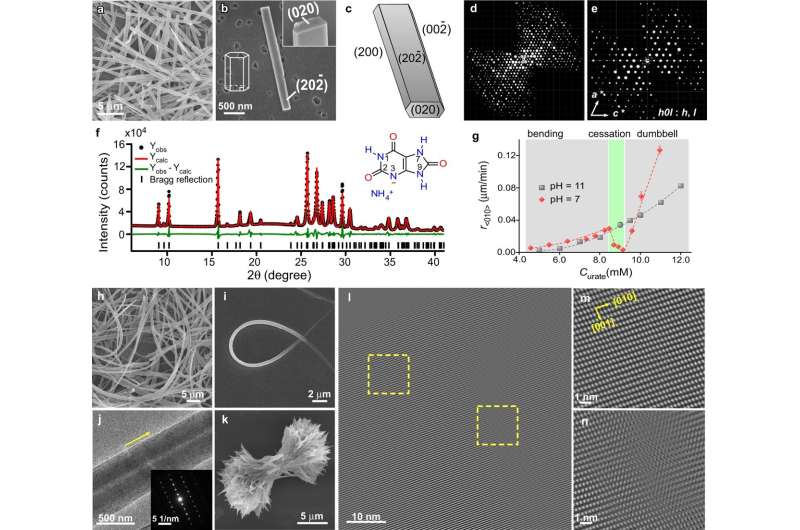
In the body, crystals—made of things such as calcium or a collection of urine—form masses that can cause pain and serious health conditions. A University of Houston crystals expert, Jeffrey Rimer, Abraham E. Dukler Professor of Chemical Engineering, known globally for his seminal breakthroughs using innovative methods to control crystals to help treat malaria and kidney stones, is reporting a new method to control the growth of ammonium urate crystals, the substance known to cause kidney stones in dolphins.
Yes, dolphins get kidney stones, too. And how did we find this out? You can thank the Navy.
In fact, move over Navy SEAL—the bottlenose dolphin is another marine mammal working hard defending our shores. With their highly evolved ability to detect objects, the dolphins have been helping the U.S. Navy find underwater mines for decades as part of the U.S. Navy Marine Mammal Program.
As part of that program, the Navy takes good care of its dolphin friends, driving research on such matters as dolphin kidney stones. Rimer has been riding the wave of dolphin research for a while, previously reporting on crystals associated with dolphin kidney stones made of ammonium urate, rarely found in humans.
Now he's leading an international team of researchers from Tianjin University China, Stockholm University Sweden, University of Pittsburgh, University of Strathclyde, Glasgow Scotland, Texas A&M University, Purdue University, Instituto Politecnico Nacional Mexico and The Molecular Foundry at Lawrence Berkeley National Laboratory.
On behalf of the team, Rimer has published new work in Nature Communications on their discovery of a method to control the crystal growth of ammonium urate by manipulating isomers of urate called tautomers. The findings may not only help dolphins but may also have implications for the pharmaceutical industry.
"We found that a small fraction of urate existing as a minor tautomer can control the rate at which crystals grow to the point they can completely block crystallization," said Rimer. "It was the most unexpected and remarkable thing to find that as you increase the concentration of urate, all of a sudden the rate of crystallization drops to almost zero and crystals do not grow in that region."
Rimer thinks it may be possible to mimic those results by diet control to get the concentration in the kidney in that range, so then the possibility exists that crystal growth would be inhibited, and medicine would be unnecessary.
That goes for dolphins and humans, alike, but more on that in future research.
Examining urate crystals, Rimer also found that tautomers get incorporated into crystals as defects, and that is where the findings have implications for pharmaceuticals.
Among the top 200 drugs, there are 33 (including allopurinol, used to treat kidney stones) that are tautomers. These medications impact millions worldwide, prescribed for HIV, epilepsy, COVID-19, schizophrenia and cancer (skin, lung and pancreatic).
"When we produced crystals with very few defects, they dissolved much slower whereas crystals with a higher percentage of defects dissolve faster," said Rimer. "That is critical for pharmaceuticals because when you put medicine into your body, their effectiveness is related to how fast they dissolve," said Rimer.
"We are asking the question about these 33 pharmaceuticals—do companies really know the extent to which they develop defects? The same question can be posed for nature where tautomers may impact unique properties in species that are vital to their intended function like optical properties in fish or color change in chameleons," he said.
With Rimer's continued research, these questions may soon be answered.
More information: Weiwei Tang et al, Tautomerism unveils a self-inhibition mechanism of crystallization, Nature Communications (2023). DOI: 10.1038/s41467-023-35924-3
Journal information: Nature Communications
Provided by University of Houston