This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers map genomic risk factors for the complex eye disease AMD
Researchers at Tel Aviv University identified a new genetic risk factor for the complex eye disease AMD (Age-related Macular Degeneration), a leading cause for loss of eyesight at an advanced age. For the first time, the researchers identified proteins that play a key role in the development and functioning of the tissue affected by the disease, found their exact sites in the genome, and discovered the connection between variations in these genomic regions and the risk for AMD.
The researchers state, "The new discovery enhances our understanding of the previously unknown function of genomic regions outside the genes. The method we applied may enable the deciphering of additional genetic mechanisms involved in various complex genetic diseases."
The study was led by Prof. Ruth Ashery-Padan and Prof. Ran Elkon and their research teams, Mazal Cohen Gulkar, Naama Mesika, Ahuvit David, and May Eshel, from the Department of Human Molecular Genetics and Biochemistry at the Sackler Faculty of Medicine and the Sagol School of Neuroscience at Tel Aviv University. The paper was published in PLOS Biology.
Prof. Ashery-Padan explains, "One of the greater challenges in genetic research today is decoding the genetic mechanisms of complex diseases caused by a combination of several different genetic and environmental factors (rather than an identifiable defect in a single gene). Diabetes, bowel diseases, and various mental illnesses are just a few examples. In our study we chose to focus on AMD, which causes degeneration of the central retina—a major cause of loss of vision at an advanced age in developed countries."
Prof. Elkon adds, "AMD has a significant genetic component. Studies comparing the genomes of people with and without AMD (as well as a range of other complex genetic diseases) have found differences in several genomic regions, probably associated with risk factors for the disease. However, these differences were not detected in any specific gene, but rather in the extensive regions that stretch between the genes, whose functions and modes of operation are still largely unknown.
"In fact, comparative studies have identified whole genomic regions that are probably related to the disease but were unable to pinpoint any specific feature in these regions and define it as a risk factor. Our study addressed this problem."
The study focused on the cells of a layer of tissue called retinal pigmented epithelium (RPE), which supports photoreceptors in the retina, and is essential for their initial development as well as their survival throughout an individual's lifetime. According to the researchers, this tissue is affected right from the earliest stages of AMD.
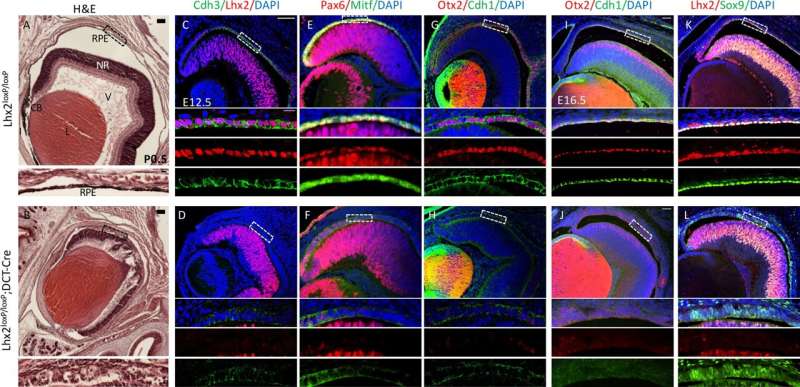
Prof. Ashery-Padan says, "First, we wanted to understand the genetic mechanism that activates and regulates the specific activity of pigmented epithelium cells. Through a series of experiments, knocking down different proteins in both a mouse model and human cells, we identified two key proteins, LHX2 and OTX2, which together dictate the expression of many genes unique to this tissue. The proteins act as transcriptional activators—binding to specific regulatory sites in the genome to determine which genes will be expressed in a particular cell."
The next challenge was mapping the precise locations of the two proteins in the genome. The researchers used the innovative technology ChIP-seq—a DNA sequencing method that identifies binding sites where proteins bind to the DNA.
Prof. Elkon states, "We found that the binding sites of the two proteins were quite close to each other. Moreover, these same sites had previously been identified as related to risk factors for AMD (namely, sequences that showed differences between people with and without AMD). We assume that due to changes in DNA sequences in these genomic regions, transcriptional proteins cannot easily find and bind with their binding sites.
"This reduces the expression of the nearby gene regulated by the transcriptional proteins, which encodes an ion channel known as important to eye function. The decrease in the gene's activity affects the entire tissue, increasing the risk for development of AMD."
Prof. Ashery-Padan summarizes, "In our study we identified two proteins related to risk factors for the complex genetic eye disease AMD. In addition, for the first time, we were able to map the exact genomic sites of these proteins and found that they operate in a region previously identified as related to risk factors for AMD.
"Our findings provide new insight into a previously unsolved issue: the functions and mode of operation of genomic sequences located outside the genes, and how they are involved in complex genetic diseases. We believe that our novel research methodology will enable the identification and mapping of many other genetic mechanisms related to AMD and other complex genetic diseases."
More information: Mazal Cohen-Gulkar et al, The LHX2-OTX2 transcriptional regulatory module controls retinal pigmented epithelium differentiation and underlies genetic risk for age-related macular degeneration, PLOS Biology (2023). DOI: 10.1371/journal.pbio.3001924
Journal information: PLoS Biology
Provided by Tel Aviv University